Delcath Systems Inc.
Scaling Liver Therapy into an Oncology Platform
Purpose of the Analysis
This isn’t about today’s valuation, it’s about owning a scalable oncology platform before the market realizes it’s no longer just a device story.
This report presents a comprehensive, long-term investment thesis on Delcath Systems Inc. The objective is not to value the business on current revenues, but to analyze the structural, technological, and strategic characteristics that may enable Delcath to evolve from a niche FDA-approved therapy into a scalable procedural platform in liver-directed oncology.
Rather than offer a short-term valuation view, this analysis investigates Delcath’s therapy platform, business model, leadership, regulatory positioning, commercial ramp, and long-term clinical expansion strategy. The intent is to frame the investment not around price targets or volatility windows, but around platform durability, procedural adoption, and strategic optionality.
Delcath represents a distinct type of opportunity, one where risk is high, but the potential rewards are nonlinear and grounded in tangible, platform-level leverage. This thesis is meant to provide the foundation for building long-term conviction.
Table of Contents
Corporate Profile
Founding & Evolution
Company Overview
Management & Leadership
Business Model & Commercial Strategy
Therapy Platform & Core Products
Indications & Pipeline Expansion Strategy
Market Opportunity
Regulatory & Reimbursement Dynamics
Competitive Positioning & Differentiation
Clinical Risk & Executional Challenges
Biotech Operating Context
Financial Profile & Capital Allocation
Long-Term Strategic Outlook
Opinion & Target View
Conclusion
Acknowledgements & Disclaimer
1. Corporate Profile
Delcath Systems Inc (NASDAQ: DCTH) is a specialty pharmaceutical company focused on developing and commercializing innovative oncology therapies, primarily through its proprietary drug delivery platform designed to target liver cancers. Their business model centers on advancing breakthrough treatments that improve efficacy while minimizing systemic side effects, aiming to address significant unmet medical needs in oncology.
By leveraging a unique technology that isolates and delivers high-dose chemotherapy directly to the liver, Delcath promises to offer new hope for patients with difficult-to-treat liver malignancies, positioning itself as a potential leader in targeted cancer therapies. For investors, the company represents a high-growth opportunity driven by clinical progress and expanding market potential within a critical segment of cancer treatment.
2. Founding & Evolution
This isn’t just a biotech turnaround, it’s the revival of a platform that’s spent two decades quietly solving one of oncology’s most intractable procedural problems.
Delcath Systems was founded with a singular goal: to transform how liver cancers are treated. Unlike traditional chemotherapy, which circulates through the entire body, Delcath aimed to deliver ultra-targeted, high-dose chemotherapy directly to the liver while sparing the rest of the body from toxic exposure.
The founding insight, that the liver’s unique vasculature could be isolated, treated, and reconnected in a single intervention, was both technically ambitious and clinically promising. It implied not just better cancer control, but potentially a better quality of life. In a field notorious for brutal trade-offs, Delcath’s proposition was simple: treat harder, harm less.
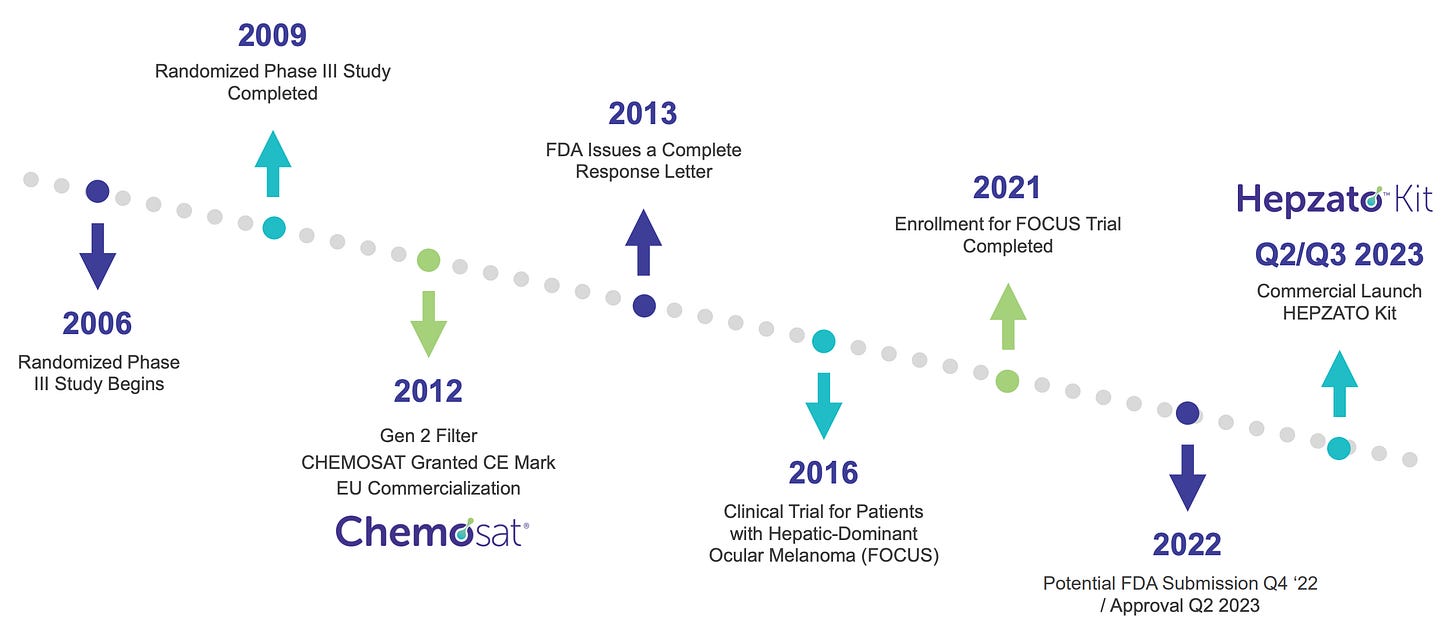
The company’s roots trace back to the late 1990s, when early prototypes of its percutaneous hepatic perfusion (PHP) system were in development. Initial years were focused on engineering a catheter-based delivery system that could perfuse the liver with high-dose melphalan while simultaneously filtering the blood to prevent systemic toxicity.
By the early 2000s, Delcath entered early clinical testing and regulatory engagement, positioning itself more as a “device-enabled pharma company” than a conventional oncology biotech. The dual identity, drug + device, would shape both its opportunities and obstacles in the years to come.
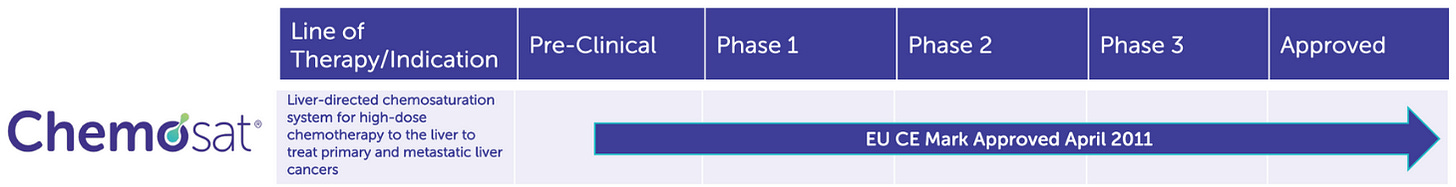
After nearly a decade of iterative development, Delcath gained European CE Mark approval in 2011 for its CHEMOSAT Hepatic Delivery System to treat liver-dominant tumors, primarily ocular melanoma metastases. But while this approval opened the door to European commercialization, it became clear that widespread adoption would be hampered by real-world hurdles: the procedure was complex, reimbursement systems were fragmented, and initial efficacy data were promising but not practice-changing.
In the U.S., the company struggled with the FDA, failing to secure approval in earlier attempts due to insufficient data and questions around safety profiles. By the mid-2010s, Delcath entered a prolonged period of retrenchment, share dilution, and strategic reassessment.
A major turning point came under the leadership of Gerard Michel, who was appointed CEO in 2020. Michel brought a pragmatic, execution-focused approach shaped by prior experience at Vericel and other healthcare companies. Under his tenure, Delcath pivoted away from pursuing narrow, niche approvals and instead set its sights on broader indications with real commercial potential.
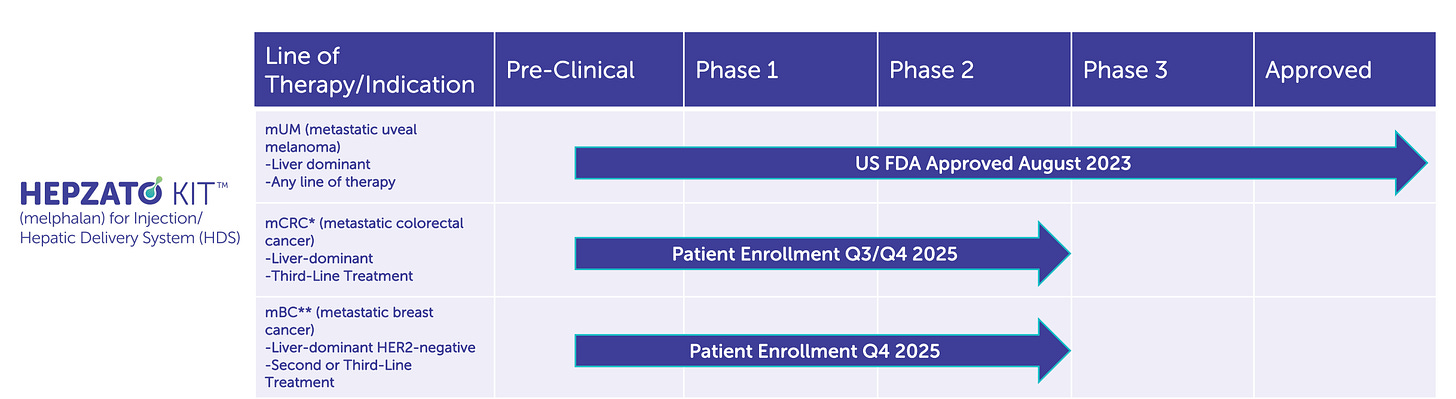
The company refined its procedure, updated safety protocols, and launched the FOCUS trial, a pivotal study targeting metastatic uveal melanoma. In August 2023, Delcath finally secured FDA approval for HEPZATO KIT (a modernized version of the PHP system), marking a long-awaited regulatory milestone. For investors, this was more than a clinical win, it was a credibility restoration.
What followed was arguably Delcath’s most important strategic shift to date: the transition from an R&D-heavy medtech platform to a commercial-stage oncology company with a viable revenue engine and future pipeline optionality.
The initial U.S. launch of HEPZATO for ocular melanoma with liver metastases is only the beginning. Delcath is now enrolling a Phase 3 trial (the ALIGN study) in colorectal cancer liver metastases, a vastly larger indication. Management’s target is clear, to create a platform that delivers liver-targeted chemotherapy across multiple solid tumors, scaling commercial infrastructure along the way.
In a biotech landscape littered with failed pivots and shallow pipelines, Delcath’s persistence is notable. Its 25-year journey has been slow, nonlinear, and often frustrating. But its progress is now compounding. With FDA approval in hand, a commercial plan underway, a next-gen delivery system expected by 2025, and clear signals of clinical utility, Delcath stands on the verge of inflecting.
The company’s history is not just one of scientific innovation, but of strategic endurance, a case study in surviving long feedback loops and emerging with a product that might finally deliver what the original vision promised.
3. Company Overview
What if one FDA-approved procedure could redefine how we treat liver metastases across multiple tumor types?
Delcath Systems occupies a unique place in oncology: a platform company whose value isn’t in molecule discovery, but in how chemotherapy is delivered. This section explores the company's therapeutic focus, product mechanism, commercial strategy, and why its procedural approach to liver tumors may unlock broader clinical and financial leverage than traditional biotech models allow.
What Delcath does: Precision chemotherapy for liver tumors
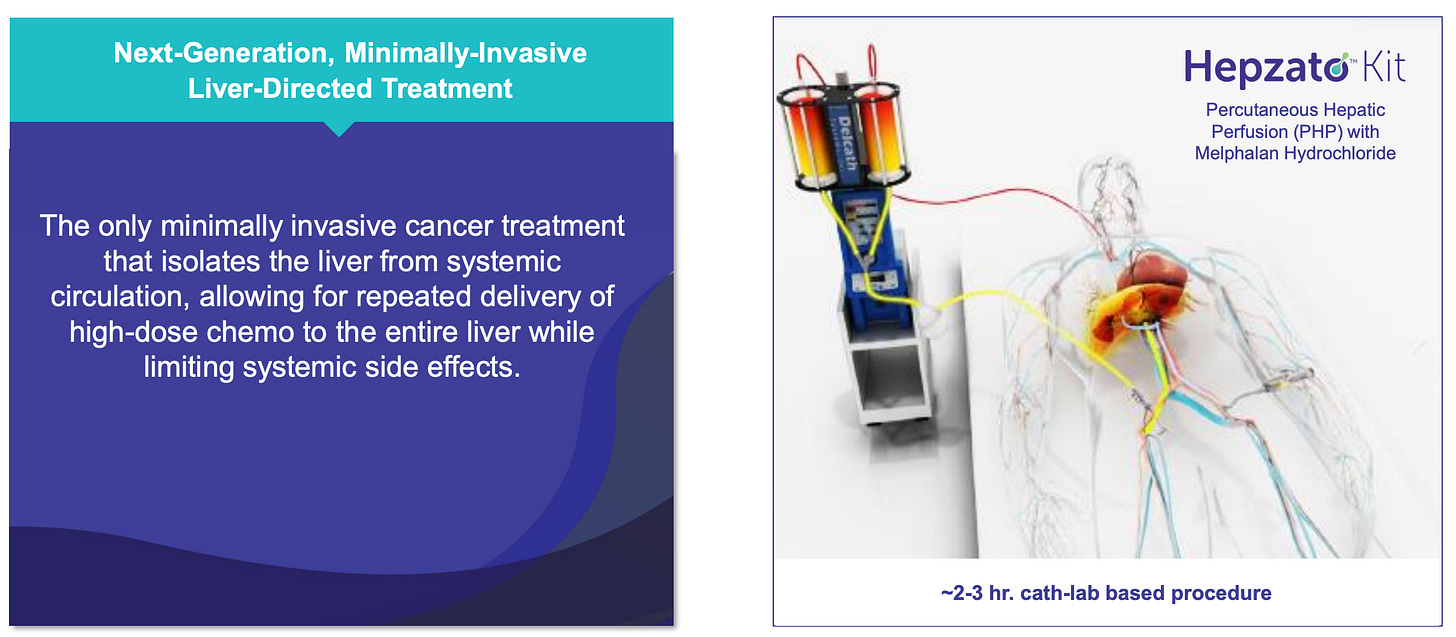
Delcath Systems Inc. is a U.S.-based oncology company that has developed and now commercializes a targeted drug delivery platform for liver-dominant cancers. Its flagship product, the HEPZATO KIT, enables physicians to deliver high-concentration chemotherapy directly to the liver, isolated from systemic circulation, through a procedure known as percutaneous hepatic perfusion (PHP). This approach allows for significantly higher drug exposure to liver tumors compared to systemic administration, while minimizing collateral toxicity to the rest of the body.
The treatment is not continuous, but episodic and repeatable, administered in a hospital setting under the supervision of interventional radiologists, oncologists, and perfusion teams. It integrates drug (melphalan), device (a dual-balloon catheter system), and filtration (extracorporeal hemofiltration) into a single therapeutic procedure. The HEPZATO KIT was approved by the FDA in August 2023 for the treatment of metastatic uveal melanoma (mUM) with liver metastases, a narrow but high-need indication that served as the company’s clinical and regulatory beachhead.
Delcath is not a traditional biotech developing a pipeline of molecules, nor is it a contract manufacturer or service provider. It operates as a procedure-driven oncology platform: a company whose core value lies in enabling hospitals to treat hepatic tumors more effectively by offering a regionally concentrated, FDA-cleared solution that is both reimbursable and clinically differentiated.
The problem: Liver-dominant metastases are a therapeutic dead zone
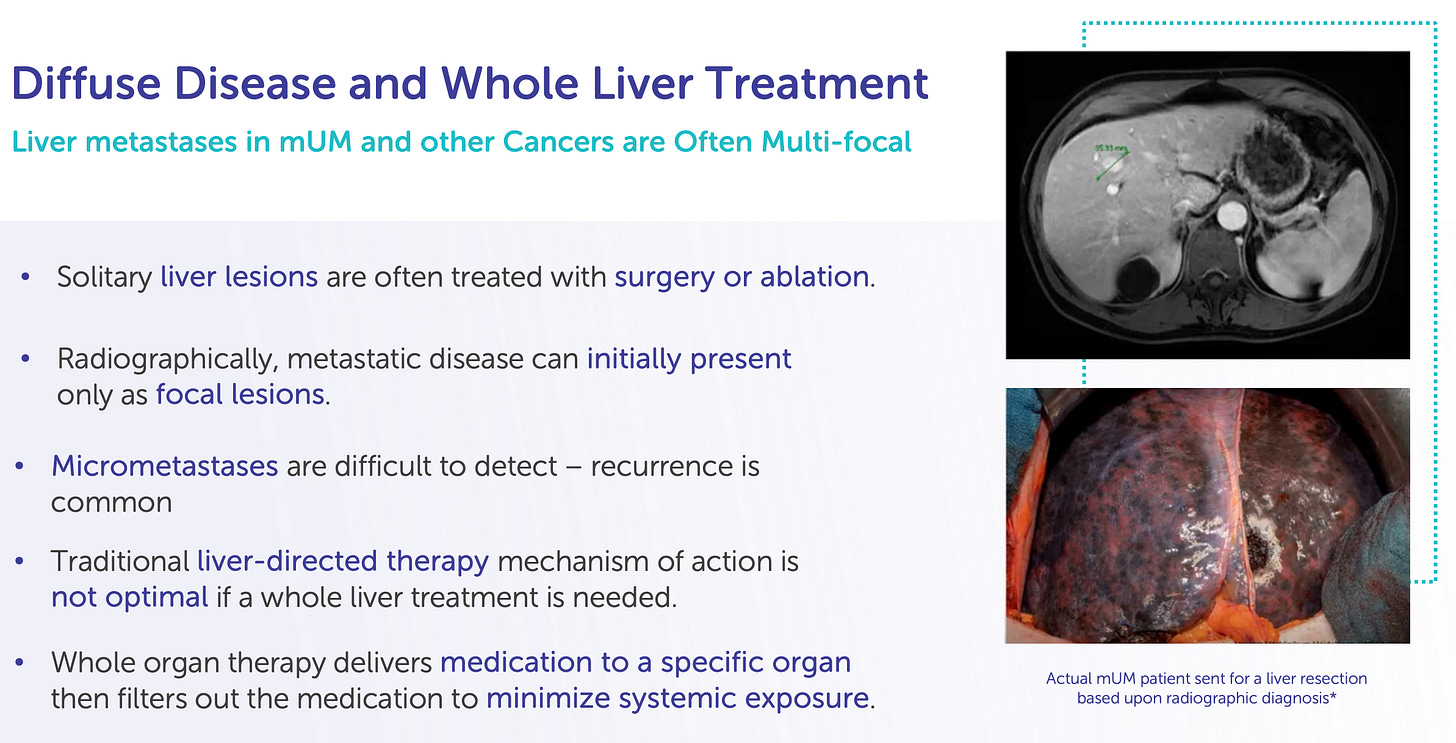
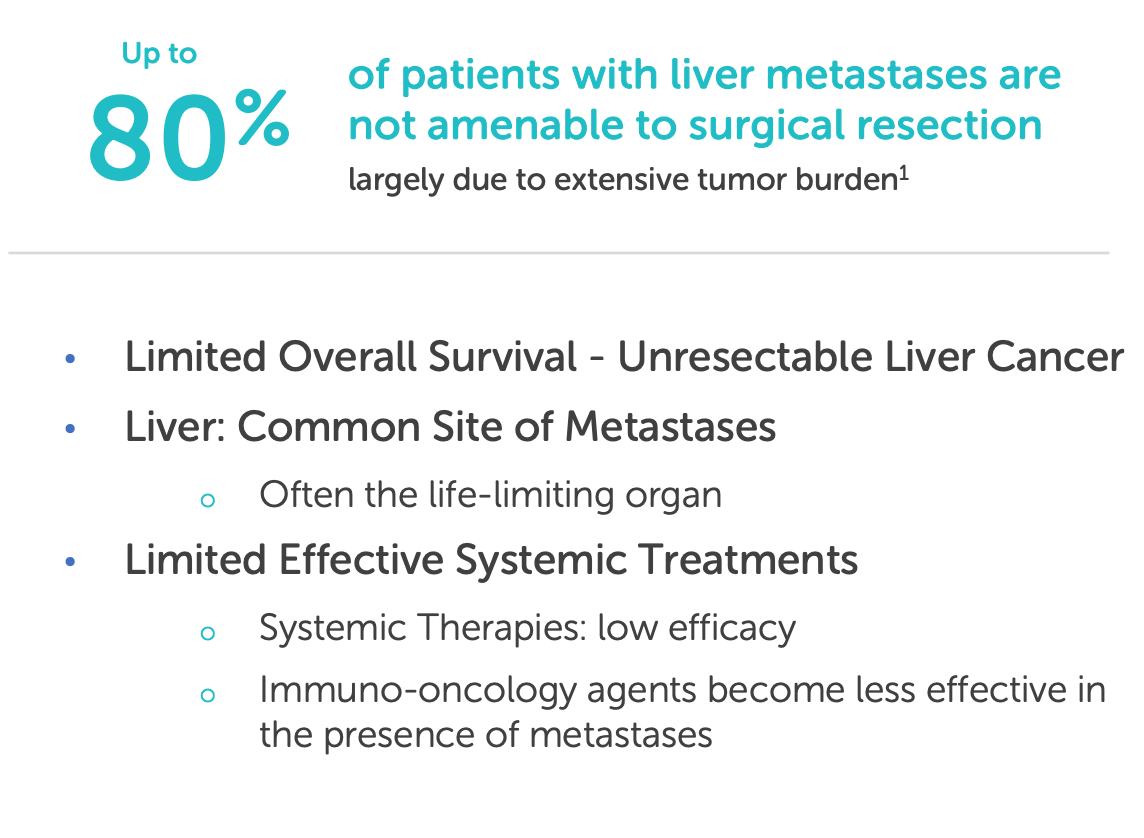
The liver is a frequent site of metastasis for multiple solid tumors, including colorectal cancer, ocular melanoma, cholangiocarcinoma, breast cancer, and pancreatic cancer. Yet systemic chemotherapy has long struggled to control liver-predominant disease due to two critical constraints: dose-limiting toxicity and heterogeneous drug delivery. Despite decades of innovation in systemic oncology, the liver remains one of the most challenging organs to treat effectively, particularly when the disease is confined to it but otherwise manageable.
For many of these patients, curative surgery is not an option, and systemic therapies offer poor hepatic control. In metastatic uveal melanoma (Delcath’s lead indication), over 90% of patients will have liver metastases as the dominant or sole site of spread, yet immune checkpoint inhibitors and small molecules have yielded limited responses. Even in colorectal cancer, where systemic options exist, patients with liver-only disease often experience hepatic progression long before other organs are affected.
The result is a mismatch between disease burden and therapeutic targeting: a liver-predominant cancer treated by whole-body drugs, often resulting in either underdosing the tumor or overdosing the patient.
The solution: A regionally targeted, systemically controlled platform
Delcath’s HEPZATO KIT is designed to solve this problem by isolating the liver during treatment, delivering a 3-5x higher localized dose of chemotherapy (melphalan), and filtering the blood before it reenters the general circulation. This is achieved via a sophisticated dual-balloon catheter that temporarily separates hepatic circulation from the systemic bloodstream, connected to an extracorporeal circuit that performs real-time hemofiltration.
The underlying mechanism is both mechanical and pharmacological: instead of discovering new drugs, Delcath is redistributing the pharmacokinetics of an existing one. Melphalan, a well-characterized alkylating agent, is used in high concentration within the liver for maximal tumoricidal activity, a practical application of “dose intensity by anatomy.” Once perfused, the drug is filtered out through a charcoal-based filter system to reduce systemic exposure. The net effect is a super-therapeutic dose to the liver, with subtherapeutic systemic levels.
This creates a unique therapeutic index: one that allows liver tumors to be targeted aggressively, while avoiding the hematologic or gastrointestinal toxicity that would otherwise occur at such doses. The technology has evolved over two decades and is now a fully integrated drug-device therapeutic platform regulated under the FDA’s NDA pathway. While the concept is not new, Delcath is the first to bring it to full regulatory and commercial fruition in the U.S. for cancer treatment.
Where Delcath stands today: Post-approval, early commercial phase
As of mid-2025, Delcath has completed the transition from a pre-revenue development-stage company to a commercial-stage oncology platform. The FDA approval of the HEPZATO KIT in August 2023, based on data from the FOCUS trial in metastatic uveal melanoma, represented the company’s first major regulatory milestone and has enabled its initial market launch in select U.S. academic and cancer centers.
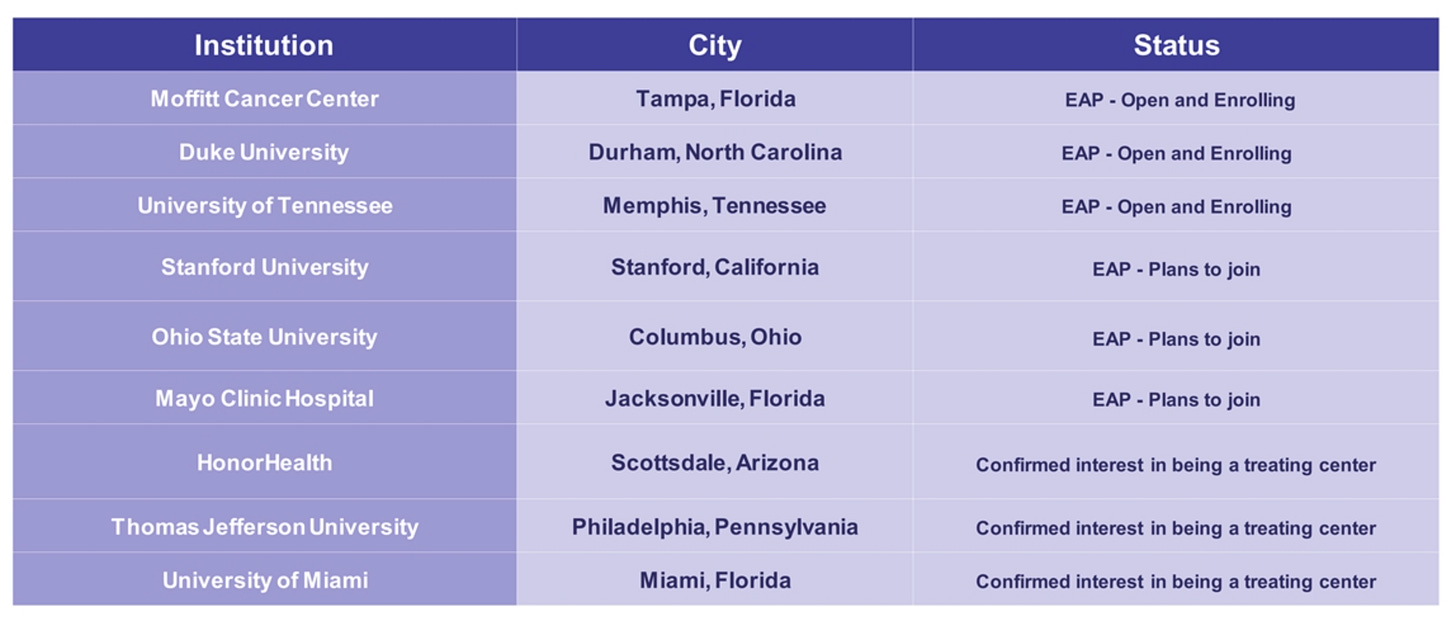
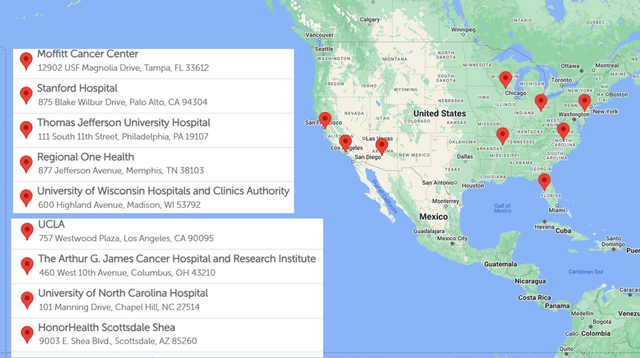
The company is now actively training physicians, onboarding institutions, navigating reimbursement dynamics, and building out field support, a classic early launch phase. Though adoption is expected to be measured given the complexity of the procedure and rare nature of the current indication, Delcath has already secured Category I CPT code assignment and is working toward broad payer coverage. Early demand signals from major liver oncology centers (e.g. MD Anderson, Memorial Sloan Kettering, Thomas Jefferson) suggest latent enthusiasm for liver-directed therapies that address gaps in current practice.
On the R&D front, the company is advancing the ALIGN Phase 3 trial in colorectal liver metastases (CRLM), which represents a vastly larger commercial opportunity. Enrollment is underway, and top-line data are expected in 2026 or early 2027. If successful, HEPZATO could become a standard of care for CRLM patients with unresectable, liver-dominant disease, a group that numbers tens of thousands annually in the U.S. alone.
Delcath has also begun development of a next-generation delivery system intended for commercial release in 2025. This upgraded kit will feature improved filtration efficiency, simplified setup, and increased operator consistency, all aimed at reducing procedural burden and expanding accessibility beyond top-tier interventional oncology centers.
At present, Delcath remains pre-profit, but is guiding toward cash flow break-even by 2027-2028, contingent on uptake in uveal melanoma, expansion into CRLM, and adoption of the next-gen kit. Cash runway and recent capital raises suggest a 2-3 year operational buffer, sufficient to navigate the commercial ramp and fund key clinical programs.
Strategic significance: A platform built for expansion, not a single indication
For long-term investors, the importance of Delcath lies not in its current revenue base, but in its positioning as a platform for liver-directed oncology. The company’s core value is its ability to deliver repeatable, liver-specific chemoperfusion across multiple tumor types and patient populations. With a single infrastructure and procedure, it can address liver metastases from colorectal, breast, pancreatic, ocular melanoma, and even primary liver tumors like iCCA and HCC, all of which share a common limitation: systemic drugs underperform in the liver.
Moreover, Delcath is entering this market at a time when interventional oncology is gaining momentum. Procedures like SIRT (Y90 radioembolization), TACE (chemoembolization), and ablation are increasingly integrated into cancer care pathways, but none offer what Delcath does: full-dose, systemically filtered chemotherapy in a repeatable, modular protocol. In this sense, Delcath is carving out a new niche that merges surgical precision with systemic pharmacology.
The timing also matters. Payers and physicians are increasingly focused on value-based oncology, therapies that meaningfully extend survival while minimizing toxicity and cost. Delcath’s therapy, though procedural, is potentially cost-saving when compared to extended systemic regimens that require months of therapy with modest hepatic control. Its product is hospital-based, high-margin, and structured for procedural billing, similar in reimbursement dynamics to SIRT or TAVR, offering durable pricing power and clinical integration.
In short, Delcath is not just a company with a device or a drug. It is a platform oncology company with a path to multiple indications, an addressable market in the billions, and a business model that, if executed properly, offers long-term revenue durability with margin expansion as procedure volumes scale.
Delcath is building more than a product, it’s constructing a procedure-based oncology platform. Its HEPZATO KIT represents the fusion of pharmacology, interventional technique, and hospital-based care, wrapped in a reimbursable, FDA-approved framework.
As the company expands indications and refines its delivery system, the core mechanism, liver-isolated chemoperfusion, remains central to its moat. For long-term investors, the real bet isn’t on one disease or product cycle. It’s on Delcath’s ability to scale a fundamentally new way of treating metastatic cancer where it lives.
Understanding the company’s strategic position sets the stage for evaluating the leadership team tasked with executing it.
4. Management & Leadership
In biotech, technology gets headlines, but leadership builds value. Delcath’s edge is execution, and its team is built to scale.
In the world of oncology platform companies, technical elegance is only as valuable as the team tasked with operationalizing it. For Delcath, the leadership bench is not just functional, it’s strategic. This section evaluates the management team not simply by credentials, but by fit: how each executive maps onto the company’s commercial, clinical, operational, and financial inflection points.
From CEO Gerard Michel’s capital discipline to Dr. Vukovic’s clinical rigor and Kevin Muir’s sales execution, Delcath’s leadership is arguably its most undervalued asset. In a sector riddled with scientific promise but executional fragility, this is a team built for precision, not just potential.
Gerard Michel - Chief Executive Officer
In biotech, especially in complex platforms that straddle device, drug, and procedure, the most important decision is often not the science , it's the steward. Executional failure, not scientific failure, is the dominant cause of value destruction in small-cap therapeutics companies. And in Delcath Systems, that execution rests on one person: Gerard Michel. He did not invent the technology, nor did he lead the early trials, but in a very real sense, he is the reason Delcath is now investable.
Michel is not a visionary scientist. He is not a capital-markets promoter. He is, by every measure, an operator, and Delcath is a company that needed exactly that. This is a story about fit: a 25-year-old platform technology finally matched with a CEO who understands what it takes to convert an elegant concept into commercial momentum. For long-term investors, Michel is not just part of the investment case, he is its operational core.
From strategy to the field: A career built on cross-disciplinary competence
Michel’s resume reads less like that of a typical biotech founder and more like that of a turnaround architect. With over 30 years of experience across biopharma, medtech, and corporate strategy, he has spent his career navigating complexity, both scientific and organizational. From early roles at Wyeth and Lederle Labs in sales and marketing, he transitioned to Booz Allen Hamilton, where he advised healthcare companies on commercial strategy and product development.
The most formative stretch of his career came in the C-suite of three mid-stage biopharma companies, NPS Pharmaceuticals, Biodel Inc., and Vericel Corporation, where he combined financial oversight with corporate development. At NPS, he helped reshape the pipeline and prepare the company for its eventual $5.2B acquisition by Shire. At Biodel, he worked through the economic and regulatory challenges of diabetes therapeutics. But it was at Vericel where Michel arguably earned his operator credentials.
At Vericel, Michel served as CFO from 2014 to 2020, where he played a central role in the company’s turnaround. He helped lead the acquisition of MACI, a marketed autologous cell therapy from Sanofi, and then restructured the company around this newly acquired commercial asset. Under his financial and operational leadership, Vericel shifted from R&D burn to revenue-generating growth, achieving break-even operations and building a high-margin niche around regenerative orthopedics. The stock rose over 1,000% during his tenure, not because of a pipeline breakthrough, but because of commercial execution and capital discipline.
The Delcath challenge: A company in search of a builder
When Michel took over Delcath in October 2020, the company had spent two decades developing a novel, but underutilized, therapy: regional chemoperfusion for liver tumors. The technology had earned CE Mark approval in Europe years earlier, and had shown promising but inconsistent results across academic centers. Yet Delcath had failed repeatedly to obtain U.S. FDA approval, burned investor trust with dilution, and suffered from a lack of clear commercial identity. It was, by most accounts, a company in search of structure, a scientific asset looking for an operator.
Michel inherited a platform with clinical data, but no market. Trials had been run but not harmonized. Payers didn’t understand the therapy. Physicians had to be trained from scratch. There was no CPT code, no formulary status, no field team, and a skeptical investor base. In short, it was a classic biotech limbo: too complex to fail outright, too fragmented to scale.
Michel brought a pragmatic re-foundation. He revised clinical strategy around a single, high-probability approval path: metastatic uveal melanoma (mUM), a rare cancer that nearly always metastasizes to the liver and lacks a systemic standard of care. This gave Delcath a clear regulatory narrative. He completed the FOCUS trial, engaged the FDA directly under the NDA pathway, and ultimately secured HEPZATO KIT approval in August 2023, the company’s first U.S. approval in over 20 years of existence.
Strategic clarity: From rare disease to procedural oncology platform
But Michel’s value to Delcath goes beyond the approval itself. His true contribution is strategic: re-framing Delcath not as a rare disease company, but as a procedural oncology platform. Under his leadership, Delcath is no longer a single-indication play. It is a system. And like all systems, it is built to scale.
The HEPZATO KIT now forms the commercial backbone. The ALIGN Phase 3 trial in colorectal liver metastases (CRLM) offers a clear expansion path into a multi-billion-dollar market. The next-generation delivery system, expected in 2025, will reduce friction at the procedure level, allowing more centers to come online. Michel is building a company that doesn't win by discovery, it wins by deployment.
This structure fits Michel’s style. He is not a biotech storyteller. In public communications, he focuses on center onboarding, CPT code integration, procedure training, and payer engagement. His earnings calls read more like operator briefings than capital raises. That sobriety has helped rebuild investor confidence in a platform many had written off.
Capital stewardship: A rare discipline in small-cap biotech
One of the most underappreciated aspects of Michel’s leadership is his capital discipline. Delcath under his tenure has avoided the kind of chronic dilution that often plagues clinical-stage biotech. In fact, despite achieving a U.S. FDA approval and initiating a Phase 3 trial, Delcath has remained lean: management headcount is modest, SG&A has scaled only in line with launch, and the company maintains tight control over cash burn. Michel has raised capital only when value inflection points justified it, and at better terms.
Moreover, Michel has built a non-promotional culture. There is no excessive use of stock-based compensation. No marketing fluff. No pie-in-the-sky projections about pan-cancer applications or AI-driven oncology. Just a clear, grounded thesis: train physicians, grow procedure volume, expand indications, improve the kit, reinvest in pipeline.
This approach is rare. In a sector where "platform" often means "perpetual pre-revenue optionality," Michel is building the opposite, a revenue-engineered system with defined economics, gross margin visibility, and a commercial moat rooted in procedural proficiency. It’s a model that may take longer, but has fewer points of failure.
Central to the thesis: Betting on execution, not just science
Ultimately, what makes Michel indispensable to the Delcath investment case is not that he believes in the platform, but that he knows how to run it. In platform therapeutics, particularly in interventional oncology, the CEO is often the business model. You’re not just betting on the science. You’re betting on a human system that can manage clinical data, payer relationships, procedure training, regulatory nuance, and logistics, simultaneously.
Delcath’s future depends on scaling a complex, high-value therapy through a fragmented and conservative U.S. hospital system. That’s not a scientific problem. It’s an execution problem. And Michel has done it before, at Vericel, at NPS, and now, step by step, at Delcath.
Investors searching for asymmetric biotech returns often chase the next molecule. But Delcath is different. The asymmetry here lies in executional inflection, the conversion of a long-languishing technology into a repeatable commercial engine. In that sense, Gerard Michel is not just a CEO. He is the de-risking mechanism for the entire investment thesis.
Executive Leadership Team
Martha S. Rook, PhD - Chief Operating Officer
Dr. Martha S. Rook brings over 25 years of technical and operational leadership spanning academic research, diagnostics, and biologics manufacturing, a background that positions her uniquely well to navigate the complexity of Delcath’s drug-device combination platform. Before joining Delcath in 2024, Dr. Rook served as Chief Technical Operations Officer at insitro, a machine learning-driven drug discovery company, where she led cross-functional responsibilities including quality systems, laboratory operations, project management, and research infrastructure.
Prior to that, she held the same title at Sigilon Therapeutics, where she oversaw the analytics, manufacturing, supply chain, and quality for a combination biologic-device product, giving her direct experience with regulatory, CMC, and logistics challenges similar to Delcath’s. Her earlier tenure at MilliporeSigma spanned 13 years, culminating in her role as Vice President and Head of Gene Editing & Novel Modalities, where she built and led a business unit supporting cell and gene therapy workflows from R&D to GMP manufacturing.
At Delcath, Dr. Rook serves as Chief Operating Officer, responsible for overseeing the company’s transition from clinical-stage to full commercial operations. Her role spans core technical functions, including manufacturing, supply chain, CMC strategy, quality systems, and commercial readiness, and she plays a pivotal role in scaling the HEPZATO KIT from a single-site launch product to a repeatable, high-throughput oncology procedure across U.S. and potentially ex-U.S. markets.
As the company prepares to roll out a next-generation delivery system in 2025, Rook is central to streamlining the procedure workflow, ensuring manufacturing robustness, and harmonizing supply chain logistics, all while maintaining the strict quality controls necessary for FDA-regulated combination products. Her operational oversight is especially critical as Delcath increases its site activation cadence and seeks to standardize treatment delivery in complex hospital settings.
Delcath’s business model depends on repeatable, procedure-based therapy delivery, a structure fundamentally different from oral drugs or infused biologics. Every treatment requires precise execution across hospital systems, making operational reliability a core source of value, not just a back-end function. In this context, the COO role becomes highly strategic. Dr. Rook’s background with combination products, supply chain scale-up, and biologic-device integration gives her direct leverage over Delcath’s ability to execute consistently across a growing network of oncology centers.
As Delcath moves toward broader market penetration, particularly with the anticipated launch of its next-generation delivery system in 2025, her leadership will be critical in reducing procedural friction, improving throughput, and ensuring that hospitals view HEPZATO not as a complex outlier but as a standardizable, system-ready intervention. For a platform whose long-term value hinges on adoption, reproducibility, and device-driven scalability, her function is not just operational support, it is core to the company’s go-to-market engine.
David Hoffman - General Counsel, Corporate Secretary & Chief Compliance Officer
David Hoffman brings more than two decades of legal and regulatory expertise in the biotechnology sector, with a particular focus on commercial-stage companies operating in highly regulated therapeutic categories. Before joining Delcath, he served as Associate General Counsel and Chief Compliance Officer at Vericel Corporation, where he oversaw all legal and compliance functions as the company transitioned from clinical stage to commercial operator in the regenerative medicine space.
His work supported the market growth of autologous cell therapies and biologics, product categories that, like Delcath’s offering, required extensive physician training, patient-specific logistics, and payer engagement. Earlier in his career, Hoffman advised a range of life sciences firms on commercial law, risk management, and regulatory structure, giving him a versatile foundation across legal domains critical to high-growth biotech companies.
At Delcath, Hoffman holds a multifunctional leadership role encompassing general counsel responsibilities, corporate governance as Secretary, and oversight of compliance as the company scales its commercial footprint. His day-to-day remit includes contract strategy, regulatory risk, intellectual property, compliance protocols, and oversight of interactions with payers, providers, and hospital systems, all essential elements of Delcath’s growth phase.
With the HEPZATO KIT now FDA-approved and commercially available in the U.S., Hoffman’s legal and compliance architecture must support site onboarding, field team operations, and interactions governed by Sunshine Act, Stark Law, and anti-kickback statutes. He also manages board governance matters and plays a key role in aligning legal operations with the company’s broader strategic, clinical, and commercial objectives.
This role is particularly important for Delcath because the company operates in a procedure-based therapeutic model, where the line between drug, device, and service can be blurry, especially in the eyes of regulators. Every site activation, reimbursement discussion, or physician training interaction carries regulatory and legal implications that require rigorous oversight.
Additionally, as Delcath prepares to expand into new indications and potentially enter ex-U.S. markets, Hoffman’s experience at Vericel, guiding a similarly complex therapy through commercialization, offers immediate strategic relevance. His presence helps de-risk Delcath’s compliance profile at a time when the company is building commercial velocity and engaging directly with hospital systems, making his role a critical enabler of scalable, audit-ready growth.
Vojislav “Vojo” Vukovic, MD, MSc, PhD - Chief Medical Officer
Dr. Vojo Vukovic brings to Delcath over two decades of senior leadership in oncology drug development, with a career spanning early- to late-stage clinical trials, regulatory engagement, medical affairs, and strategic indication planning. His experience is both broad and deep: he has served as Chief Medical Officer or Senior Vice President of Clinical Development at multiple oncology-focused biotechs including Aileron Therapeutics, Taiho Oncology, and Synta Pharmaceuticals, where he led the advancement of various novel therapies across solid tumors and hematologic malignancies.
His academic background includes an MD from the University of Sarajevo, followed by an MSc in radiation biology and a PhD in tumor biology from the University of Toronto, a training path that has given him scientific fluency across both mechanistic oncology and translational development. A frequent contributor to oncology literature and active member of major professional societies including ASCO, AACR, ASH, and ESMO, Dr. Vukovic brings with him a respected voice in the cancer research community and a track record of global clinical leadership.
As Chief Medical Officer at Delcath, Dr. Vukovic is responsible for overseeing all clinical development activities, including trial design, regulatory strategy, medical monitoring, and physician engagement, as well as long-term indication planning for the HEPZATO platform. He leads the medical strategy behind the ongoing ALIGN Phase 3 trial in colorectal liver metastases (CRLM) and is a key architect of the clinical rationale for expanding the platform into other liver-dominant cancers such as intrahepatic cholangiocarcinoma (iCCA), hepatocellular carcinoma (HCC), and potentially breast and pancreatic metastases.
In addition to his role in clinical trial execution, he plays an important cross-functional role in medical affairs, helping to shape physician education, investigator-initiated studies, and treatment center engagement, all of which are essential to building procedural adoption in the interventional oncology community.
For Delcath, the CMO role is more than clinical trial oversight, it’s about narrative leadership and clinical franchise building. The company’s platform relies not just on FDA approvals, but on sustained evidence generation across tumor types and procedural standardization across institutions.
Dr. Vukovic’s ability to communicate the hepatic-directed treatment rationale to both regulators and practicing oncologists is key to driving indication expansion and ensuring that each clinical program is designed for regulatory clarity, payer relevance, and physician uptake. In a company where commercial growth will depend on real-world clinical traction and a multi-indication label strategy, Dr. Vukovic’s presence provides scientific credibility and strategic rigor, making him a vital pillar in Delcath’s long-term oncology roadmap.
Kevin Muir - General Manager, Interventional Oncology
Kevin Muir brings over 20 years of sales and marketing leadership in the biotherapeutics and interventional medtech sectors, with deep domain expertise in commercializing complex, procedure-based therapies. Prior to joining Delcath, he served as Director of U.S. Sales for the Interventional Oncology business at BTG, where he was instrumental in the market success of TheraSphere, a Yttrium-90 (Y90) radioembolization platform used in the treatment of liver cancer.
TheraSphere, like Delcath’s HEPZATO KIT, required physician education, site onboarding, logistical coordination, and payer alignment, giving Muir direct operational experience in navigating the commercial hurdles of liver-directed, minimally invasive oncology interventions. His earlier roles included Director of Sales at ClearFlow Inc. and Aragon Surgical, as well as leadership positions at Kensey Nash, Kyphon, and Genzyme Biosurgery, where he built regional and national sales organizations targeting both hospital-based and outpatient markets.
At Delcath, Muir leads all commercial operations related to the HEPZATO KIT and future platform therapies, with a specific focus on hospital engagement, sales force development, and interventional oncology center activation. He is tasked with building the field team responsible for training, adoption, and procedural throughput, ensuring that institutions not only purchase the therapy, but are equipped to administer it effectively.
His remit includes center of excellence development, physician relationship management, sales enablement infrastructure, and feedback loops with the clinical and operations teams to inform rollout priorities. Given the procedural nature of HEPZATO, which requires cross-functional coordination across radiology, oncology, perfusion, and pharmacy, Muir’s role demands more than typical oncology drug commercialization. He must establish an ecosystem of repeat users, internal champions, and reimbursement pathways that can support durable product utilization.
This role is mission-critical for Delcath because the company’s revenue is procedure-based and site-driven, not broadly prescribed. Unlike small molecule oncology drugs, HEPZATO must be adopted hospital by hospital, and every additional treatment center represents a significant revenue opportunity and fixed-cost leverage point.
Muir’s specific experience at BTG scaling TheraSphere, which shares nearly identical clinical dynamics in hepatic oncology, gives him a precise playbook for growing Delcath’s footprint. His blend of medtech fluency, sales force leadership, and oncology commercialization makes him central to Delcath’s near-term inflection and long-term value realization. As adoption expands and the next-generation delivery system approaches, Muir will be responsible for turning a de-risked platform into a scalable, durable commercial franchise.
Sandra Pennell - Chief Financial Officer
Sandra Pennell brings over two decades of senior financial leadership across high-growth life sciences companies, with deep expertise in SEC reporting, capital markets execution, FP&A, and treasury operations. Her career includes leading finance organizations through periods of clinical development, public market volatility, and commercial scale-up, all highly relevant to Delcath’s current trajectory.
Prior to joining Delcath, Pennell served as Vice President of Finance at Invivyd, Inc., a publicly traded infectious disease therapeutics company, where she helped manage equity financing, regulatory filings, and operational budgeting in a rapidly evolving clinical landscape. Before that, she held the role of Vice President & Corporate Controller and Principal Accounting Officer at Vericel Corporation, where she played a key role during the company’s transformation from R&D-centric to a profitable commercial-stage enterprise.
At Delcath, Pennell is responsible for all finance and accounting functions, including financial reporting, audit and compliance, internal controls, capital structure strategy, investor relations support, and strategic resource allocation. She plays a cross-functional role in budgeting for the commercial rollout of HEPZATO, guiding investment decisions across sales force expansion, clinical trial resourcing, and next-gen platform development.
Importantly, she oversees the alignment of Delcath’s cost structure with its staged revenue plan, ensuring that operational scale-up remains capital-disciplined and ROI-focused. Her involvement in treasury and equity financing strategy positions her as a key interface with capital markets, responsible for maintaining the financial runway needed to execute the company’s commercial and clinical agenda without over-reliance on dilutive capital raises.
The CFO function is especially pivotal at Delcath given its position at the intersection of capital-intensive operations and high-margin procedure-based revenue. Unlike pure-play biotech models that live between inflection points, Delcath’s long-term viability depends on disciplined, sustained investment in both commercial and manufacturing infrastructure, without bloating SG&A ahead of revenue scale.
Pennell’s background at Vericel, which shares a similar trajectory as a once-early-stage therapeutic company that matured into a commercial platform, offers a valuable operational blueprint. Her approach to financial planning and risk management is essential to guiding Delcath through its transition phase, managing dilution risk, and ensuring that every capital deployment aligns with strategic growth levers. As Delcath moves into multi-indication expansion, Pennell will serve as the financial architect ensuring scalability, compliance, and capital efficiency across the platform lifecycle.
Delcath’s platform is complex, its market fragmented, and its therapeutic model demands precision across domains most biotechs never have to coordinate: device regulation, procedural training, hospital reimbursement, and real-world treatment execution. What sets Delcath apart is not just its innovation, but the architecture of the team tasked with scaling it.
From top to bottom, the company’s leadership reflects a rare coherence: operators with experience commercializing procedure-based therapeutics, managing regulatory ambiguity, and driving adoption one hospital at a time. For long-term investors, this is not a management team to tolerate, it is a strategic moat in its own right.
With the leadership in place, the next step is to assess how Delcath plans to translate its platform into a scalable, durable business.
5. Business Model & Commercial Strategy
This isn’t a pill you prescribe, it’s a platform you build. Delcath’s business model doesn’t follow biotech rules, and that’s exactly the point.
Delcath Systems is not a conventional biotech company monetizing molecular innovation through prescriptions or out-licensing. Instead, it operates a procedure-centric business model, in which revenue is generated each time a hospital administers its flagship therapeutic product, the HEPZATO KIT, as part of a structured interventional oncology treatment.
This model aligns more closely with medical device companies like Intuitive Surgical or Medtronic, or procedural therapeutic platforms like SIRT (Y90 embolization) or TAVR (valve replacement), than with traditional oncology drug developers.
The implications for investors are material: Delcath has the potential to scale high-margin, repeatable revenue by expanding treatment center access, increasing per-site utilization, and broadening its indication footprint, all while maintaining capital discipline and avoiding the linear SG&A ramp typically associated with multi-product pharmaceutical companies.
What Delcath sells: A high-value procedure packaged as a drug-device combination
At the center of Delcath’s business model is the HEPZATO KIT, an FDA-approved, proprietary system designed to administer high-dose melphalan hydrochloride directly to the liver via isolated perfusion, while simultaneously filtering the patient’s blood before it returns to systemic circulation. The system is classified as a drug-device combination product, comprising several integrated components:
A dual-balloon catheter system to isolate hepatic blood flow,
A precision-controlled extracorporeal hemofiltration circuit,
A high-concentration chemotherapy dose of melphalan,
Procedural disposables and supporting materials tailored for safe, repeatable use.
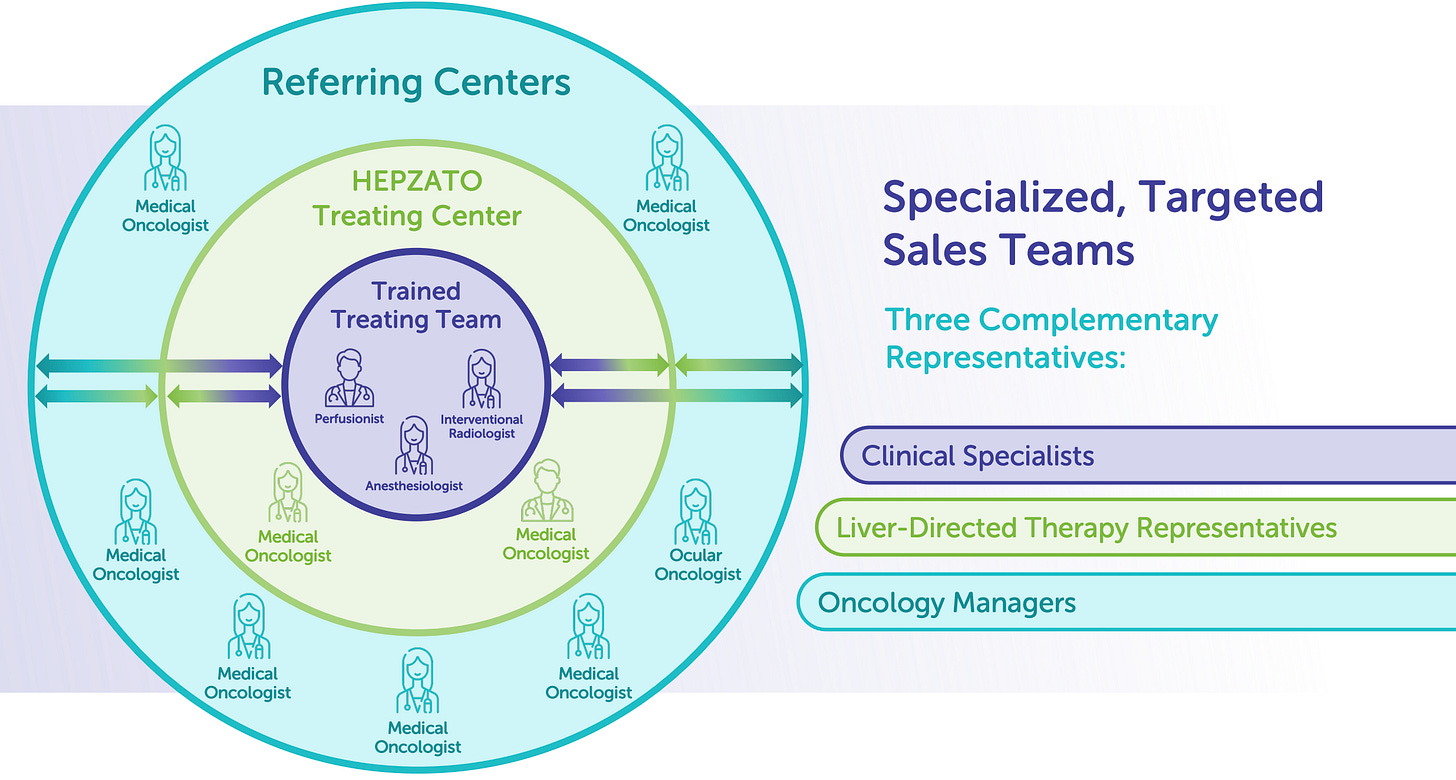
The product is delivered as a complete, single-use kit intended to be used during a structured interventional oncology procedure. This therapy is not administered through conventional pharmacy channels or as an outpatient infusion, it is delivered within a hospital’s interventional suite, typically by a multi-disciplinary team involving an interventional radiologist, perfusionist, anesthesiologist, and oncology staff.
Because the therapy is episodic, with most patients undergoing 2-4 treatment cycles per clinical protocol (or more in some cases), Delcath monetizes each procedure independently. That translates into multiple revenue events per patient and a built-in element of semi-recurring revenue within each treatment center. Importantly, the complexity of the procedure creates clinical and operational lock-in, once a center is trained, it is unlikely to churn or substitute another therapy, especially as procedural repetition improves staff efficiency and cost recovery.
How Delcath makes money: High ASPs, repeat utilization, and site-level revenue density
Delcath sells the HEPZATO KIT directly to hospitals and academic centers through specialty distribution or direct contracting, with the average selling price (ASP) estimated between $25,000 and $35,000 per procedure. The ASP reflects not only the therapeutic value of melphalan, but also the complexity and IP protection surrounding the delivery system. Gross margins are expected to exceed 70% at scale, consistent with high-end oncology procedures and minimally invasive interventional therapies.
The revenue model is tightly tied to site activation and patient throughput, not broad prescriber volume. This means the company's top-line growth is dependent on the number of operational centers and their procedural cadence, not traditional metrics like market share among oncologists or script volume. Once a site is trained and credentialed, it becomes a revenue-generating asset. Centers treating patients with hepatic-dominant cancers often have multiple eligible patients at any given time, enabling fast conversion from launch to recurring use.
Patients typically receive multiple treatments within a defined window, so per-patient lifetime value is high, particularly when considering the limited competition in mUM and future planned indications. Delcath also benefits from favorable economic incentives for hospitals, many of which seek to expand their interventional oncology service lines with procedures that are both clinically meaningful and reimbursable. HEPZATO offers both, and the company supports procedural adoption with clinical training, onsite technical support, and reimbursement navigation.
The business model allows for high revenue density per center. As sites gain experience, procedural cadence often increases, enabling more efficient staff utilization and consistent revenue streams. Early commercial rollout is focused on high-volume academic centers, where Delcath can maximize revenue per activation while building procedural champions that will aid in community center adoption over time.
Platform leverage: Multi-indication expansion without commercial duplication
Delcath’s most strategic commercial advantage lies in its platform leverage, the ability to expand into new indications without rebuilding its commercial, technical, or procedural infrastructure. Unlike small molecule drugs that require new salesforces, educational campaigns, or specialty pharmacies for every new label, HEPZATO is a modular procedure.
Once a hospital is trained to administer the therapy, that same team, catheter infrastructure, and billing protocol can be used across multiple indications, whether treating uveal melanoma, colorectal liver metastases, intrahepatic cholangiocarcinoma, or eventually hepatocellular carcinoma.
This gives the company an attractive non-linear growth curve: future trials and indications expand revenue not by opening new markets from scratch, but by deepening utilization within already-onboarded centers. For example, the ongoing ALIGN Phase 3 study in CRLM, which targets a 10-20x larger market than mUM, could unlock dramatically more revenue from existing centers that have already cleared the initial onboarding and training hurdle. Importantly, Delcath does not need to scale its SG&A in parallel, it can add new patient segments without replicating the cost structure.
This also contributes to the company’s procedural moat. Because physicians, administrators, and support staff invest time and resources to integrate HEPZATO into their practice, they are less likely to switch to alternatives, even if future competition enters the hepatic-directed therapy space. The company benefits from institutional inertia, procedural repetition, and clinical familiarity, all of which build network effects around each treatment center.
From a long-term strategy perspective, this model positions Delcath not just as a single-product company, but as a procedural oncology platform with multiple verticals and optionalities. As more data emerges, and as new indications are approved, the company can stack utilization across tumor types, enhancing both per-site economics and enterprise value without proportional increases in headcount or infrastructure.
Capital efficiency, monetization timeline, and strategic alignment
Delcath’s business model is not only margin-accretive and procedurally anchored, it’s also designed for time-sensitive monetization with capital-efficient scalability. The typical onboarding process for a new treatment center takes between 3 to 6 months, encompassing contracting, physician and staff training, and initial procedural support. Once live, these sites can begin administering HEPZATO immediately, creating a relatively fast conversion from site activation to cash-generating revenue.
Hospitals purchase the HEPZATO KIT either via direct sales agreements or through specialty distributors, and Delcath recognizes revenue upon shipment, simplifying working capital management. While individual contract structures can vary by institution, the pricing model is generally based on per-use kit purchases, not volume-based tiering or rebates, which helps preserve ASP integrity in the early launch phase.
Over time, Delcath expects this structure to support robust unit economics, especially as procedural frequency rises and operational support becomes more standardized. Importantly, Delcath is not building out a large field force to “push” product, instead, it is selectively targeting high-volume academic centers where procedural oncology infrastructure already exists, allowing each site to reach meaningful revenue contribution without extensive commercial lift.
Because each new indication can reuse the same sales infrastructure and clinical support pathways, Delcath gains multi-indication leverage, every new label expansion increases throughput per center, improving fixed cost absorption and expanding gross margin. Additionally, the company’s next-generation delivery system, expected in 2025, is designed to simplify procedural setup, shorten training cycles, and reduce variability, all of which should further compress onboarding time and increase per-site utilization, reinforcing both scalability and time-to-revenue compression.
In the long-term, this structure creates a business model with multi-layered operating leverage: (1) fixed costs amortized across growing procedural volume, (2) recurring use within centers as new patients enter treatment, and (3) platform stacking via additional indications using the same delivery hardware and clinical team.
When combined with a reimbursement architecture aligned to existing CPT codes and hospital workflows, the result is a commercial engine that scales predictably, supports high-margin growth, and aligns tightly with long-term investor interests. For a small-cap company transitioning from R&D to commercialization, this business model, anchored in procedure-driven, site-recurring revenue, provides a foundation not only for top-line expansion, but also for durable margin growth and platform-level strategic optionality.
Delcath’s business model is not just innovative, it’s structurally advantaged. By anchoring revenue to procedure volume rather than prescription flow, the company builds durable, high-margin, and site-recurring income with limited commercial overhead. Each new treatment center becomes a revenue node, each new indication a throughput multiplier.
With embedded training, procedural complexity, and reimbursement alignment, Delcath creates economic and operational stickiness that is difficult to displace. And because the platform is modular, expansion doesn’t require parallel cost scaling, it deepens utilization within the same footprint. For long-term investors, this isn’t a typical biotech curve. It’s a platform economics story, and one with real-world friction that translates into real-world defensibility.
To understand how value is captured, we must now examine the underlying products and platform driving that value.
6. Therapy Platform & Core Products
Delcath isn’t delivering a drug, it’s delivering control. Precision, protection, and platform scalability are built into every procedure.
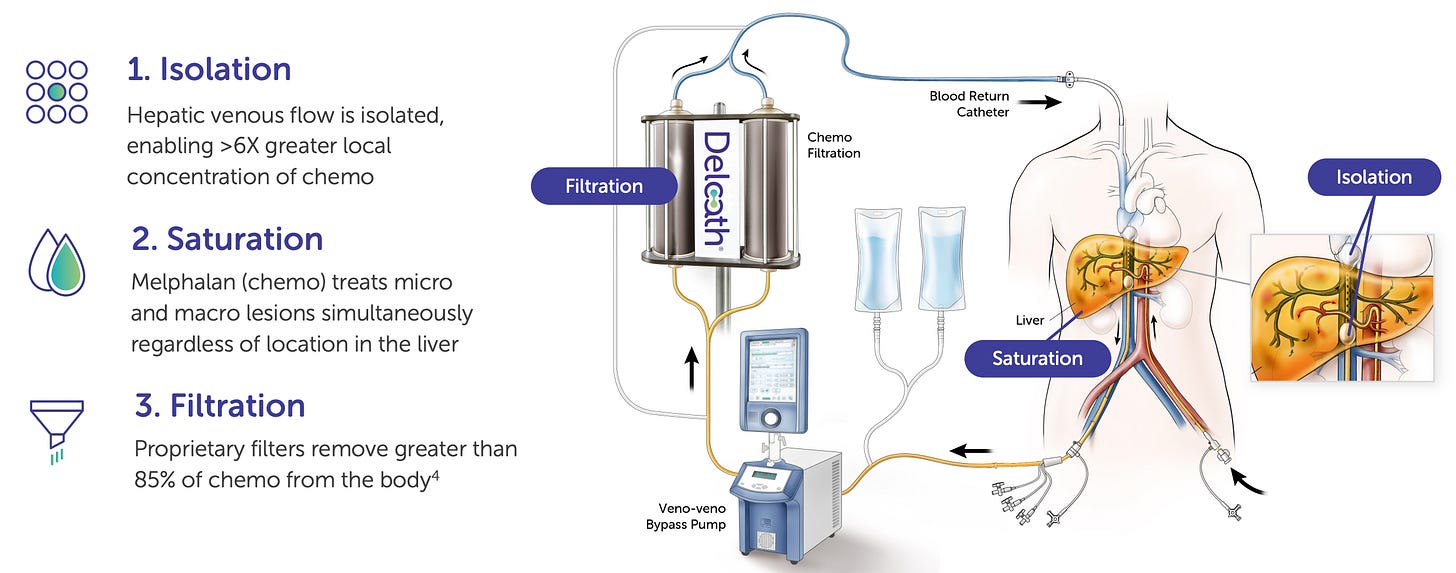
Delcath’s product innovation centers around the HEPZATO KIT, a proprietary, FDA-approved drug-device combination therapy designed for regionally targeted delivery of high-dose chemotherapy to the liver. The product is not a drug in the conventional sense, nor is it a standalone device; rather, it is a highly engineered procedural system that integrates pharmacology, interventional radiology, and extracorporeal filtration into a unified clinical solution.
The therapeutic rationale is straightforward: by isolating the liver’s circulatory system and perfusing it directly with concentrated melphalan, physicians can achieve tumoricidal drug levels within the organ while minimizing systemic toxicity through blood filtration, a pharmacokinetic profile that cannot be replicated with systemic administration.
At the product level, the HEPZATO KIT includes:
A dual-balloon catheter system inserted via the femoral artery and vein, enabling blood flow isolation of the hepatic circuit.
A high-dose melphalan hydrochloride formulation optimized for intra-arterial infusion.
A custom extracorporeal filtration circuit, which removes 85-90% of the drug before blood re-enters systemic circulation.
Procedure-specific disposables (tubing, connectors, pumps) integrated into an operating kit that supports a single use.
A mobile, table-side CHEMOSAT system, in some markets, that supports circuit management (EU only).
The delivery system is designed to be used in hospital interventional oncology suites under general anesthesia, with procedural support from perfusionists, interventional radiologists, anesthesiologists, and oncology staff. Each treatment session typically lasts several hours and is conducted on an inpatient or outpatient basis depending on institutional protocol. Patients may undergo multiple sessions, usually spaced 6-8 weeks apart.
The mechanism: Pharmacokinetic engineering through mechanical isolation
The therapeutic logic behind the HEPZATO KIT lies in its ability to mechanically reshape the pharmacokinetics of chemotherapy. At its core, Delcath’s system enables regional isolation of the liver’s blood supply, allowing for the direct infusion of high-dose melphalan hydrochloride into hepatic circulation, followed by extracorporeal filtration of the blood before re-entry into systemic circulation. This addresses one of oncology’s oldest and most persistent tradeoffs: delivering a drug dose potent enough to kill tumors without exceeding systemic toxicity thresholds.
Unlike traditional chemotherapy regimens, where drug levels are diluted across the entire body and limited by systemic side effects, Delcath’s platform concentrates exposure where the disease resides, the liver, while removing excess drug before it circulates systemically.
Published pharmacokinetic data shows that hepatic drug exposure is increased by as much as 20-fold relative to systemic delivery, while systemic drug concentrations are reduced by 85-90% due to the filtration circuit. This allows for both higher efficacy at the site of disease and an acceptable safety profile, particularly in cancers where hepatic progression is the dominant cause of mortality.
From an engineering standpoint, the key innovation lies in how the system achieves this isolation and filtration in a repeatable, reproducible clinical setting. A dual-balloon catheter blocks the hepatic venous outflow while the arterial supply is cannulated for drug infusion. Blood is routed through a proprietary hemofiltration circuit, which uses a high-capacity charcoal filter to remove melphalan before blood is returned to the body via a separate catheter.
This entire process is controlled in real-time by a trained perfusionist and requires anesthesia, hemodynamic monitoring, and coordination across interventional radiology and oncology teams, a complexity that acts as both a barrier to entry and a durable procedural moat.
Crucially, this mechanism does not require a molecular breakthrough, it requires precise control of anatomy, circulation, and filtration, which Delcath has engineered into a commercially viable therapeutic workflow. The result is a form of regional chemotherapy intensification that cannot be achieved through conventional infusion methods, positioning HEPZATO as a unique and differentiated therapeutic approach within the interventional oncology landscape.
Product evolution: From device platform to integrated combination product
Delcath’s product strategy has evolved from a hardware-centric device offering (CHEMOSAT) to a fully integrated drug-device combination product (HEPZATO). This transition reflects both a maturation of the platform and a strategic decision to pursue higher regulatory defensibility, stronger economic capture, and improved clinical usability.
Originally introduced in Europe under the CE mark as a device-only system, CHEMOSAT required hospitals to source melphalan separately, which complicated logistics and limited Delcath’s pricing power and brand control. While the platform saw clinical uptake in Europe, the lack of drug integration and FDA clearance constrained commercial scale.
Recognizing these limitations, Delcath reoriented its U.S. strategy around a combination product regulatory pathway, leading to the development of the HEPZATO KIT, a packaged system that includes both the drug and device components under a unified label. This repositioning brought several strategic advantages:
It allowed for control over drug supply, quality, and labeling, ensuring consistency across procedures.
It enabled Delcath to pursue FDA approval under a New Drug Application (NDA), gaining regulatory exclusivity and a stronger IP position.
It simplified hospital adoption by reducing friction in procurement, pharmacy prep, and procedural planning.
The FDA approval of the HEPZATO KIT in August 2023 for patients with metastatic uveal melanoma marked the platform’s first U.S. commercial milestone. This version reflects multiple product refinements: catheter design improvements, streamlined disposables packaging, more standardized operator protocols, and integration with hospital procedural workflows. These changes reduce training time, procedural variability, and error risk, all essential for driving adoption in busy hospital environments.
Looking ahead, Delcath is developing a next-generation version of the delivery system, targeting a 2025 release. While details are limited, investor communications and product roadmaps suggest upgrades may include:
Automated pressure and flow monitoring to reduce operator burden.
Simplified circuit priming and setup, improving procedural turnaround.
Potential software integration for procedural guidance and analytics.
The intent is clear: to make HEPZATO more scalable, less manually intensive, and ready for broader adoption, including community-based oncology centers. In doing so, Delcath isn’t just improving usability, it’s enhancing the long-term operating leverage and install-base ROI of every treatment center onboarded.
IP, manufacturing, and product defensibility
Delcath’s product moat is protected by a hybrid of intellectual property, regulatory exclusivity, and procedural complexity, a structure that creates real barriers to entry and supports premium pricing. On the IP front, the company holds patents covering the catheter design, hemofiltration media, methods of use, and drug-device integration, many of which extend into the 2030s.
The combination product designation adds a further layer of protection, as any generic entrant would need to demonstrate equivalence not only in drug formulation but in procedural performance and filtration kinetics, a formidable hurdle, especially given the system’s engineering-specific clinical claims.
But the defensibility extends beyond IP. HEPZATO is fundamentally a procedural solution, not a passive drug or device. Its effective use requires specialized training, cross-disciplinary coordination, and infrastructure integration. Hospitals that invest in onboarding, including perfusion staffing, protocol approval, and pharmacy integration, build operational commitment that is not easily unwound. This creates a sticky install base, similar to surgical platforms or radiation systems, where switching costs and institutional inertia reinforce long-term utilization.
From a manufacturing standpoint, Delcath uses a contract manufacturing model, outsourcing production of both drug and device components while maintaining control over final kit assembly, packaging, and quality assurance. This approach limits capital intensity while allowing for scalable production as volume ramps.
As the company moves toward its next-generation system, opportunities for cost-of-goods optimization and logistics simplification are likely to improve gross margins, an important lever as revenue scales and margin expansion becomes a core driver of equity value.
The combination of proprietary components, clinical workflow lock-in, and regulatory complexity means that even with future competition in liver-directed therapy, Delcath is structurally advantaged as the first mover with an FDA-approved, clinically validated system. In oncology, where time-to-market, institutional trust, and procedural standardization are paramount, this type of integrated defensibility can support a decade or more of uncontested commercial runway.
Strategic significance: A scalable procedure, not a one-off product
HEPZATO’s importance to Delcath’s investment thesis goes well beyond its current approval in metastatic uveal melanoma. It is the cornerstone of a procedural oncology platform designed for multi-indication expansion. Because the therapy is delivered through the same physical infrastructure and clinical workflow regardless of tumor type, each newly approved indication can be layered onto existing sites with minimal additional training or cost. This makes the platform economically and operationally scalable, a key point of differentiation from traditional biopharma models.
For example, if a hospital is treating mUM patients with HEPZATO today, that same center could treat patients with colorectal liver metastases, intrahepatic cholangiocarcinoma, or hepatocellular carcinoma using the same catheter systems, filtration setup, perfusion staff, and billing pathways. This stacking of indications creates non-linear revenue growth, as per-site utilization increases without requiring new salesforce expansion, major product reconfiguration, or commercial reinvestment. As a result, HEPZATO is not a static SKU, it is a delivery franchise capable of addressing multiple high-value, high-burden hepatic cancers.
This structure enables Delcath to pursue a stepwise platform rollout, where each clinical and regulatory milestone builds upon the last, increasing ROI on both R&D and commercial infrastructure. It also positions the company favorably within hospital systems increasingly focused on procedural consistency, training efficiency, and bundled care pathways. The more indications that can be treated through a single procedural protocol, the more attractive the platform becomes for institutional adoption and payer integration.
Over time, the HEPZATO architecture could also support future innovations beyond melphalan. While the company’s current focus is on expanding indications for its existing therapy, the mechanical isolation and filtration principles could, in theory, be applied to other agents or even combination regimens. That optionality isn’t priced into current operations but represents a strategic long-tail for long-term investors betting on platform-level oncology infrastructure.
HEPZATO is more than Delcath’s first product, it’s the chassis for an entire procedural oncology platform. By engineering therapeutic intensity into a repeatable system, the company has created a franchise that can scale across cancers, institutions, and indications with minimal reinvention. Every new use case becomes a force multiplier, not a dilution of focus.
With strong IP, built-in clinical complexity, and high procedural stickiness, the platform is both protected and extensible. In a market dominated by molecules, Delcath offers something rarer: a mechanistically sound, operationally scalable, and economically durable approach to cancer therapy.
With the platform defined, attention turns to how it expands across clinical indications and market segments.
7. Indications & Pipeline Expansion Strategy
Delcath’s pipeline isn’t about new products, it’s about deepening value from the same platform, one indication at a time.
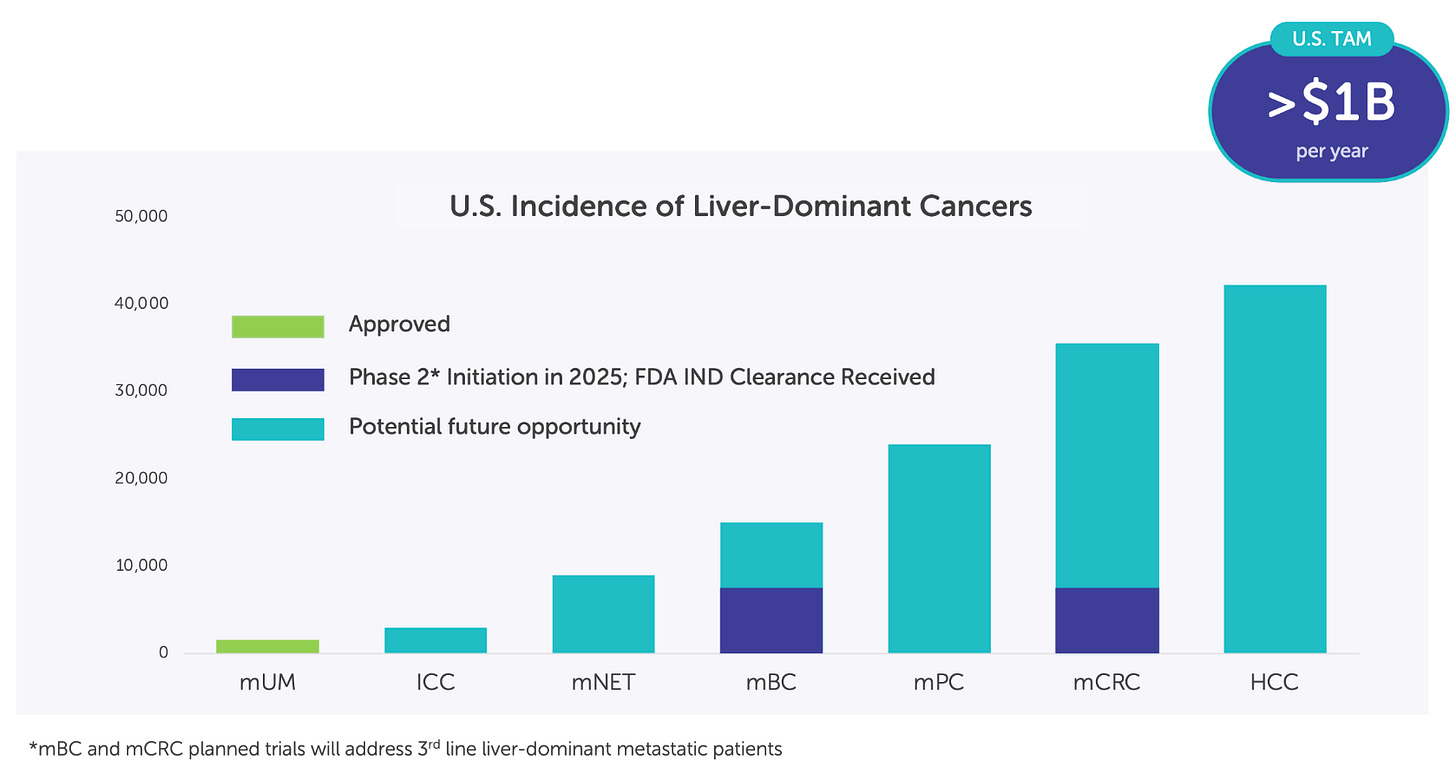
Delcath’s clinical and commercial strategy centers around the strategic layering of liver-dominant cancer indications onto a single procedural platform. With HEPZATO’s approval in metastatic uveal melanoma (mUM) as the foundational step, the company is executing a multi-year, indication-led expansion strategy targeting other high-burden, underserved liver cancers, including colorectal cancer liver metastases (CRLM), intrahepatic cholangiocarcinoma (iCCA), and hepatocellular carcinoma (HCC).
Each indication leverages the same treatment system, procedural workflow, and commercial footprint, enabling the company to scale utilization and revenue without proportionally scaling cost or infrastructure.
The clinical rationale across these indications is unified: in each case, the liver serves as the dominant site of progression and often the proximate cause of death. However, unlike solid tumors where systemic therapy can offer durable control, liver-dominant disease is frequently non-curable, poorly responsive to chemotherapy, and ineligible for resection or ablation. Delcath’s value proposition lies in its ability to deliver potent, liver-targeted cytotoxic exposure, addressing a clear therapeutic gap with precision and repeatability.
The sequencing of these indications is not opportunistic, it is engineered. Each step is meant to incrementally validate the platform across increasingly prevalent disease settings, growing the TAM while building procedural muscle, payer comfort, and clinical evidence. In this section, we explore the strategic and clinical logic of each target tumor type, its commercial relevance, and its role in the platform’s evolution.
Metastatic Uveal Melanoma (mUM): Foundational proof of concept and controlled market entry
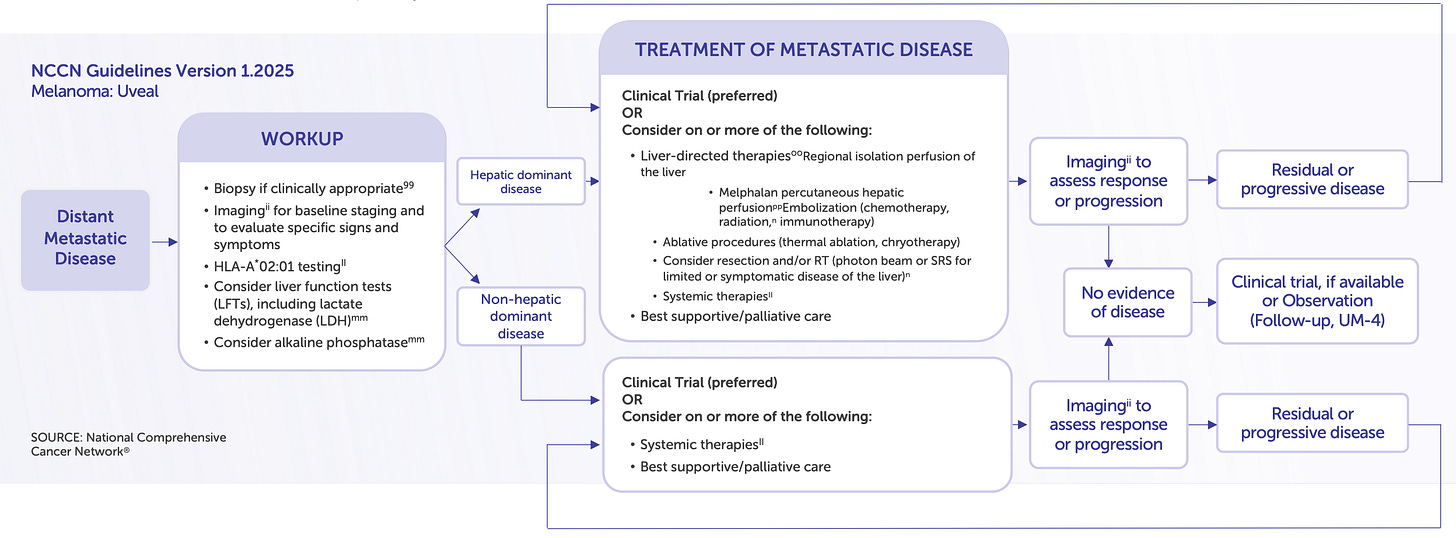
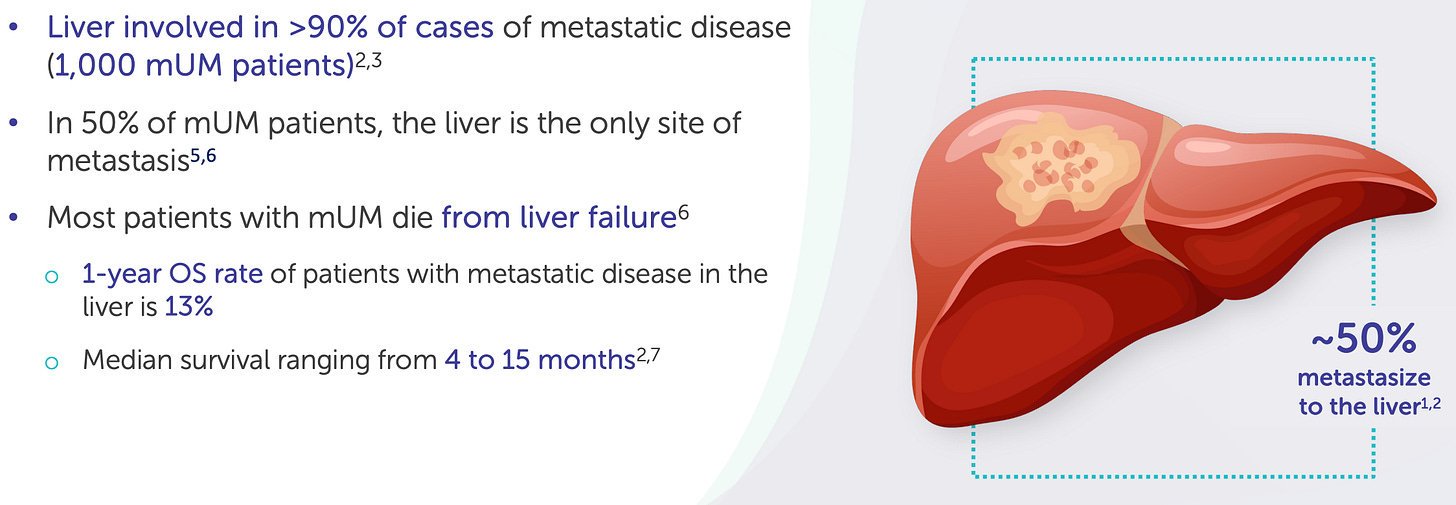
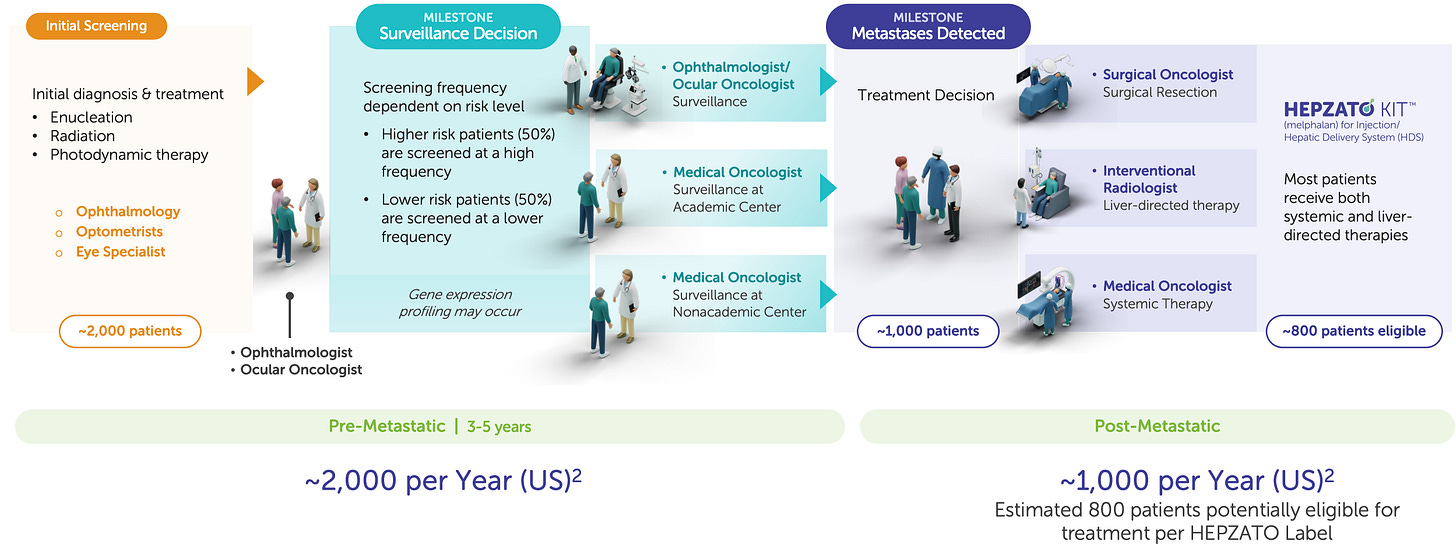
Delcath’s first FDA-approved indication, metastatic uveal melanoma (mUM), is more than a regulatory milestone; it is a strategic test bed for platform deployment, commercial refinement, and procedural adoption. With approximately 1,000–1,200 new cases per year in the U.S., mUM represents a small but highly concentrated oncology market, with most patients referred to a limited number of academic centers specializing in rare ocular cancers. More than 90% of metastatic spread occurs in the liver, making mUM uniquely suitable for a liver-directed therapy such as HEPZATO.
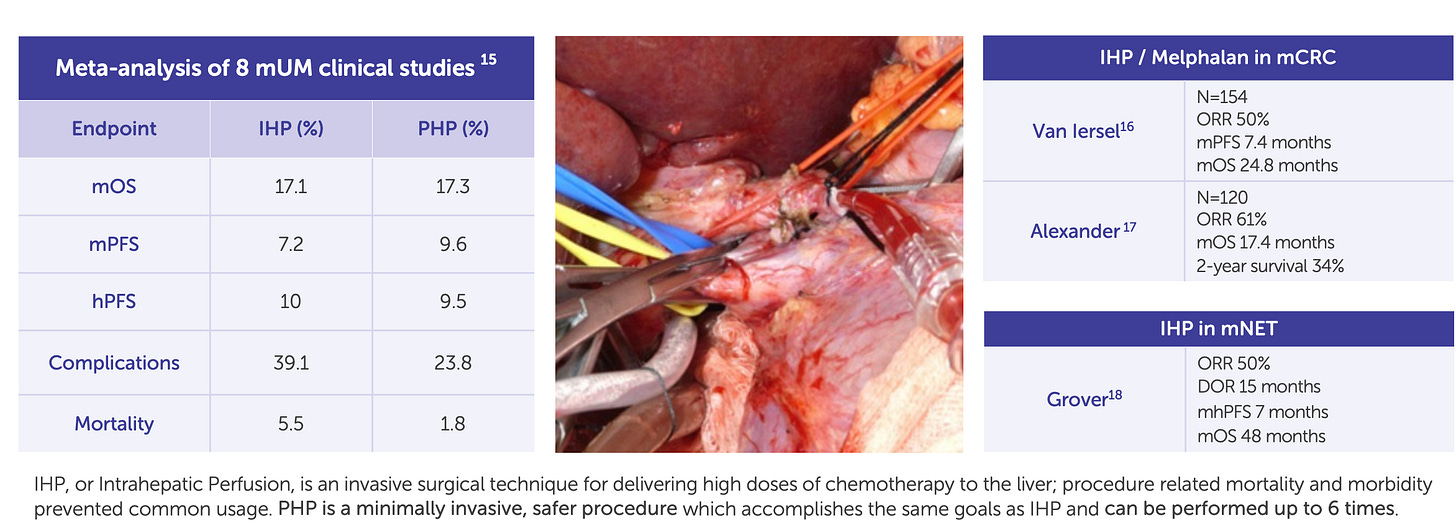
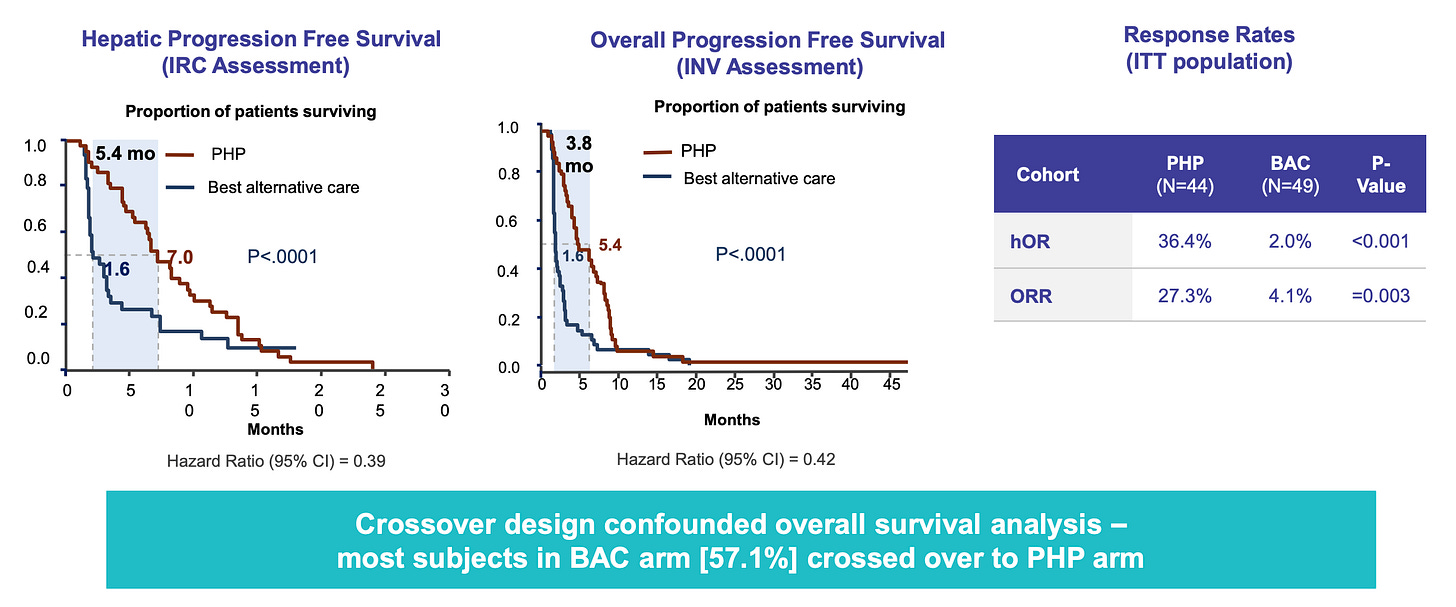
Delcath’s Phase 3 FOCUS trial (single-arm, multi-center) demonstrated that HEPZATO achieved a 36.3% objective response rate (ORR), a disease control rate of 73.8%, and a median hepatic progression-free survival (hPFS) of 8.1 months. These results were achieved in heavily pre-treated patients with no other liver-directed therapeutic options and minimal response to systemic checkpoint inhibitors. The FDA approved the HEPZATO KIT in August 2023, marking the first time a percutaneous hepatic perfusion (PHP) system was approved as a drug-device combination product for solid tumor treatment in the U.S.
From a commercial perspective, mUM allows Delcath to refine its go-to-market model in a low-volume, high-specialization setting. Most patients are funneled to major academic centers, reducing sales force requirements and concentrating onboarding efforts. The procedures are reimbursed under existing hospital frameworks, and the rarity of the disease often justifies institutional investment in specialized treatments.
Importantly, mUM centers frequently treat other complex solid tumors, particularly metastatic colorectal and cholangiocarcinoma, which means that early procedural investments are reusable across future indications.
Also, mUM provides the company with initial revenue visibility, clinical validation, and payer touchpoints that will support broader expansion. The importance of mUM lies not in its market size but in its strategic optionality, it serves as a regulatory foothold, procedural pilot, and reputational wedge into a market that values clinical differentiation.
Colorectal Liver Metastases (CRLM): The pivotal growth catalyst
If mUM was Delcath’s foot in the door, colorectal cancer liver metastases (CRLM) represent the key to unlocking platform-scale commercial potential. Colorectal cancer is the second-leading cause of cancer death in the U.S., and approximately 50% of patients eventually develop liver metastases. Among these, a large subset present with unresectable, liver-dominant disease that cannot be treated with surgery or ablation, and who have already progressed on standard chemotherapeutic or targeted regimens. In this setting, there is currently no liver-directed therapy with demonstrated survival benefit, creating a significant unmet need that Delcath is well-positioned to address.
The company’s ongoing Phase 3 ALIGN trial is enrolling patients with third-line liver-dominant CRLM. The study is designed to compare HEPZATO KIT to physician’s choice of best alternative care (typically FOLFIRI, trifluridine-tipiracil, or regorafenib), using hepatic progression-free survival (hPFS) as the primary endpoint and overall response rate and overall survival as key secondaries. Notably, the trial is powered to detect clinically meaningful differences in hepatic disease control, which has emerged as a validated proxy for overall outcomes in this patient population.
From a platform economics perspective, CRLM is transformative. The U.S. addressable population for third-line liver-dominant CRLM is estimated at 10,000-15,000 patients annually, an order of magnitude larger than mUM. Moreover, treatment sites for CRLM are often the same academic centers already activated for mUM, allowing Delcath to layer additional revenue per center without requiring incremental field buildout. Even modest penetration in this indication could support triple-digit revenue growth, with attractive gross margins and minimal SG&A expansion.
Beyond the numbers, CRLM validates the broad utility of the platform. If Delcath can show that its system improves liver disease control in one of the largest solid tumor indications, it will fundamentally reposition the company from an orphan-disease niche player to a mainstream procedural oncology business. The top-line readout is expected in 2026, making this trial the single most important clinical catalyst for the platform in the next 24-36 months.
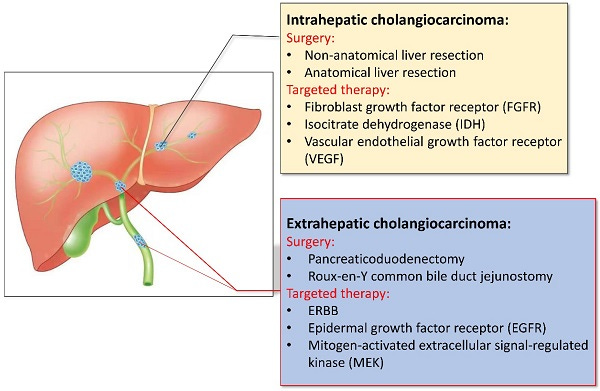
HCC and iCCA: Optionality-rich expansion into broader hepatic oncology
Following CRLM, Delcath is targeting expansion into intrahepatic cholangiocarcinoma (iCCA) and hepatocellular carcinoma (HCC), two aggressive primary liver cancers where local progression drives morbidity and mortality. Both diseases are characterized by late-stage diagnosis, limited surgical candidacy, and suboptimal response to current systemic therapies.
While agents such as durvalumab (in HCC) and FGFR inhibitors (in iCCA) have modestly improved outcomes in subsets of patients, local control remains a major challenge, especially when tumors are refractory or confined to hepatic lobes.
In HCC, the challenge is greater: checkpoint inhibitors and tyrosine kinase inhibitors (TKIs) are now standard of care, but only a subset of patients experience durable response. Delcath’s system could theoretically be used in early-intervention, locoregional control, or as a second-line approach for patients with partial response.
However, the presence of competing liver-directed modalities (e.g. TACE, Y-90) and a more complex systemic-treatment backdrop make HCC a higher-risk, higher-uncertainty target. For this reason, Delcath appears to be approaching HCC cautiously, with trial timing and design yet to be finalized.
For iCCA, Delcath is currently designing a clinical development strategy that may begin with a Phase 2 basket trial, focusing on patients with unresectable, liver-confined disease who have failed first-line gemcitabine–cisplatin regimens.
Given the small patient population and lack of direct procedural competitors, iCCA represents a clinically high-need, low-competition indication that could be brought to market with relatively low trial cost and operational complexity. Preliminary feasibility studies, along with prior experience in similar perfusion protocols, suggest strong clinical rationale.
Strategically, both iCCA and HCC extend the clinical relevance and commercial breadth of the platform. Importantly, they do so without requiring any hardware changes. The same HEPZATO catheter, filtration system, and procedural setup can be used for all indications, making these programs essentially incremental revenue unlocks rather than new product lines. Over time, they also enhance Delcath’s positioning as a go-to platform for hepatic oncology interventions, a space with limited innovation and high hospital willingness to adopt specialized procedures.
Strategic indication sequencing: Platform economics by design
Delcath’s indication sequencing is not simply a function of clinical feasibility, it is a carefully staged platform development plan, designed to optimize regulatory de-risking, commercial compounding, and capital efficiency. Each step along the indication roadmap is chosen for its ability to validate new aspects of the platform while supporting increased utilization of existing infrastructure.
mUM proves that the system can be safely implemented, reimbursed, and scaled in a real-world setting.
CRLM is the commercial breakout, the first large-market test of platform utility and payer viability.
iCCA/HCC extend the platform horizontally into additional tumor types and broaden the clinical utility of the system.
Future potential expansions (e.g., breast, NETs, or pancreatic liver metastases) offer long-tail optionality without platform redesign.
This sequencing creates a compounding flywheel: each new indication increases site-level ROI, improves revenue per procedure suite, and deepens Delcath’s procedural moat. It also allows for reimbursement harmonization across tumor types, enhancing payer relationships and supporting the case for bundled or platform-based contracting models in the future.
From an investor’s standpoint, this structure enables a rare combination of clinical development leverage and commercial reusability, every R&D dollar has the potential to expand multiple revenue lines, and every new label expansion increases throughput without increasing commercial overhead. In a capital-constrained biotech environment, this stackable ROI profile is a distinguishing strength.
Delcath’s expansion strategy is not about breadth for its own sake, it’s about compounding utility across a stable, high-barrier platform. With each new indication, the company is not just unlocking more patients, it’s increasing the throughput and economic density of every center it serves.
The result is a rare biotech profile: capital-efficient growth, infrastructure reuse, and procedural entrenchment that deepens over time. This is not a pipeline of molecules, it’s a pipeline of leverage. And if the clinical data holds, Delcath won’t need to rebuild itself for each new market. It will just need to keep the system running.
Each new indication adds strategic weight, but it only matters if the market opportunity is large and accessible.
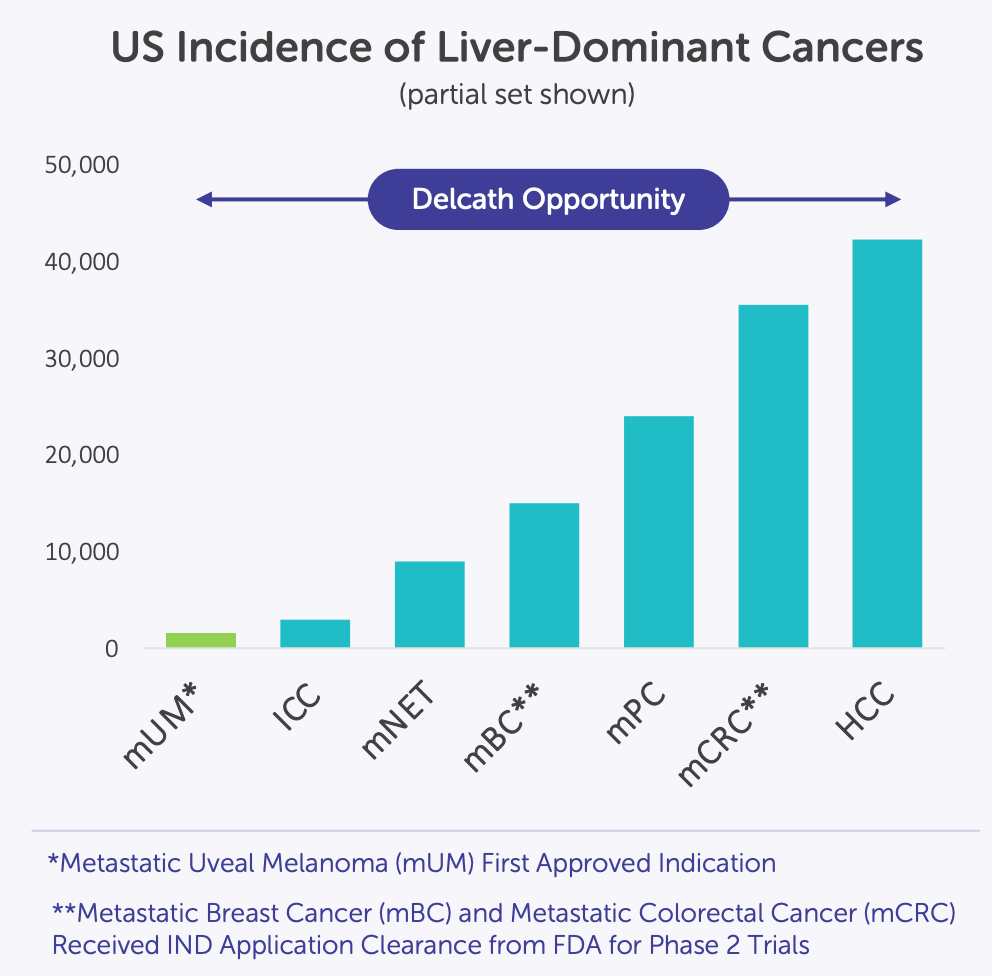
8. Market Opportunity
Delcath isn’t chasing tumor types, it’s building a procedural monopoly on the liver, one indication at a time.
To understand Delcath’s commercial potential, you have to think anatomically, not categorically. This is not a company going after discrete tumor types with separate pipelines, it’s a platform designed to dominate a single organ that plays a central role in the progression of countless solid tumors: the liver.
By anchoring its model in a repeatable, liver-specific therapy, Delcath is turning a fragmented oncology market into a unified procedural opportunity. This section breaks down how the company’s TAM is constructed not by chasing diseases, but by treating the place where they converge.
A liver-centric view of oncology: Why Delcath’s addressable market is structurally unique
The liver is one of the most frequent and consequential sites of metastatic spread for solid tumors. It is also the primary origin for some of the most aggressive cancers globally. This biological reality creates a uniquely centralized opportunity for Delcath: a therapy platform focused entirely on liver-directed oncology. The company’s proposition is not limited to a single tumor type, but rather to a shared clinical condition, liver-dominant disease, that occurs across many oncologic settings.
What makes this attractive from an investment standpoint is the procedural unification of a fragmented oncologic burden. Delcath is not developing separate solutions for each cancer subtype; it is building a system that targets the liver as a disease nexus, regardless of the primary tumor.
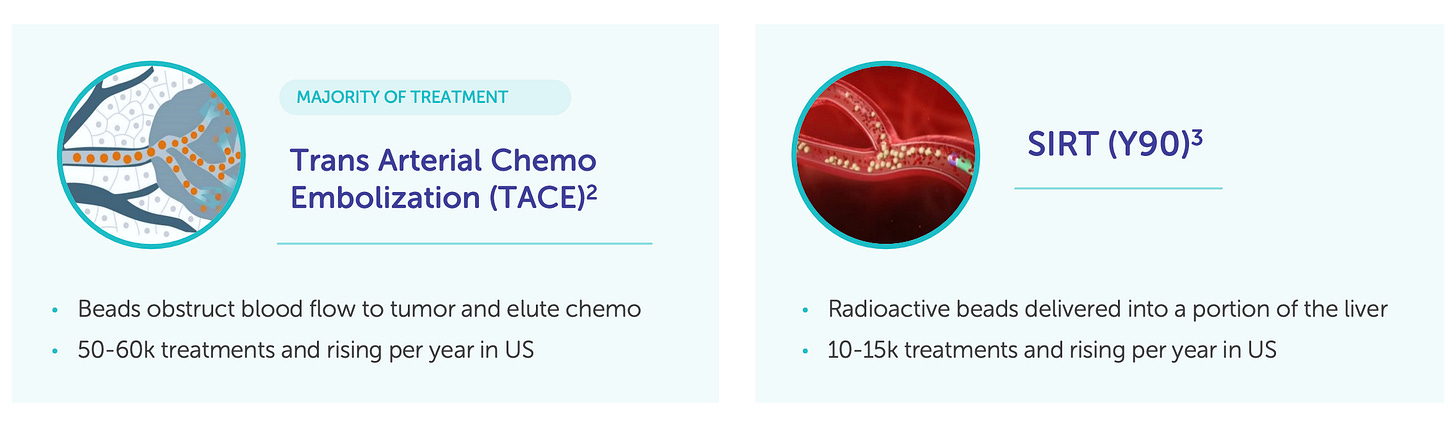
This anatomical focus creates a structurally unique addressable market. Solid tumor metastases to the liver are common in colorectal, breast, pancreatic, gastric, neuroendocrine, and ocular cancers. In fact, in colorectal cancer alone, more than 50% of patients develop liver metastases. Despite this, therapeutic solutions specifically optimized for liver control are extremely limited.
Conventional systemic chemotherapies have poor hepatic targeting and are often discontinued due to systemic toxicity. Local therapies such as transarterial chemoembolization (TACE) and radioembolization (Y-90) exist, but are either limited to certain anatomical distributions or restricted by patient liver function.
Delcath’s HEPZATO KIT is unique in that it isolates the liver via percutaneous filtration, allowing for high-dose regional chemotherapy with minimal systemic exposure. This leverages the anatomical reality of liver progression to create a therapeutic category of its own, one where the company can build broad relevance across multiple indications without needing to retool its platform.
This liver-centric strategy reframes how TAM is defined. Delcath is not chasing rare cancers; it’s organizing its development around a common organ of progression and capturing oncology market share through a verticalized procedural approach. The total market isn’t a sum of separate indications, it’s a stackable, overlapping population unified by anatomical vulnerability.
Market segmentation: Quantifying the initial and expansion indications
Delcath’s addressable market unfolds in a sequence: starting with a rare disease (mUM), then expanding into more prevalent and commercially impactful indications. Each indication brings a distinct epidemiological footprint, reimbursement profile, and regulatory risk. Together, they offer a layered, strategically staged TAM.
Metastatic Uveal Melanoma (mUM): Roughly 1,000–1,200 patients per year in the U.S. progress to hepatic metastases. These patients often have no effective treatment options, especially outside the HLA-A*02:01 genotype eligible for tebentafusp. mUM is rare but concentrated in top-tier academic centers, making it ideal for platform onboarding and procedural learning curves.
Colorectal Cancer Liver Metastases (CRLM): This is the commercial backbone of the expansion plan. Approximately 150,000 new colorectal cancer cases are diagnosed in the U.S. each year. Of these, over 50% will develop liver metastases during the disease course. Delcath targets the third-line liver-dominant segment, estimated at 10,000–15,000 patients annually. This group has failed prior systemic therapy and remains dominated by hepatic progression, making them prime candidates for liver-directed therapy.
Intrahepatic Cholangiocarcinoma (iCCA): Around 8,000–10,000 patients per year are diagnosed in the U.S., many with unresectable tumors confined to the liver. Standard of care is gemcitabine/cisplatin, but median survival remains poor. Delcath’s approach could offer a bridge to disease control in patients who are still liver-confined after first-line failure.
Hepatocellular Carcinoma (HCC): The largest of the expansion indications in global terms. While systemic therapies (e.g., durvalumab + bevacizumab) dominate first-line, liver-directed approaches remain critical in certain subgroups: those with limited response, contraindications to systemic agents, or localized failure. Estimated U.S. market of 10,000-15,000 candidates for liver-directed salvage therapy.
Adding these together, the U.S. market across these core indications approaches 30,000-40,000 patients per year, many of whom would be eligible for multiple HEPZATO treatments. With pricing estimated at $25,000-$35,000 per procedure and 2-4 treatments per patient, the revenue potential per indication is significant, even at modest penetration rates.
Revenue per patient, per site, and over time: A multi-level monetization model
Delcath’s revenue model is built not on volume drug sales or short-course therapy pricing, but on a multi-tiered monetization structure rooted in procedural adoption, repeat treatment, and durable site utilization. This creates several distinct levers of revenue growth that can scale together, a key differentiator in biotech models where revenue is often linear and tied directly to prescription volumes.
At the patient level, revenue is generated through the sale of the HEPZATO KIT, a drug-device combination that includes melphalan hydrochloride for injection and the proprietary percutaneous hepatic perfusion (PHP) system. Each kit is designed for a single procedure, and patients typically receive between 2 and 4 procedures, depending on treatment response and clinical guidelines.
With a per-procedure ASP estimated between $25,000 and $35,000, revenue per patient could range from $50,000 to $140,000, excluding hospital facility fees and services, which are separately reimbursed. The multi-treatment nature of HEPZATO, combined with its localized delivery and tolerability, makes this repeat-use structure clinically rational and economically scalable.
On a per-site basis, monetization is even more compelling. Because the HEPZATO system requires hospital training, credentialing, equipment integration, and coordination across interventional radiology, oncology, and anesthesiology, each activated site represents a semi-permanent commercial channel.
These are not “one-time” accounts; they are recurring revenue nodes. Once operational, a center may treat 5-10 patients per month, particularly as new indications become available. Assuming 3 procedures per patient and an ASP of $30,000, a single site operating at this volume could generate $5 million to $10 million annually in kit sales alone.
Moreover, this revenue does not require proportional increases in SG&A. Delcath’s salesforce is not selling to individual prescribers but rather engaging hospital systems, IR teams, and cancer centers. The commercial relationship is procedural, not transactional, which allows revenue per rep and per account to scale far more efficiently than in conventional oncology commercialization. With proper reimbursement, each incremental patient and procedure adds high-margin revenue with minimal incremental cost, driving operating leverage and capital efficiency as the business grows.
Lastly, this model benefits from stackable site monetization: as new indications (CRLM, iCCA, HCC) come online, the same hospitals treating mUM patients can expand their procedural volumes without requiring retraining or new infrastructure. This transforms each center from a static revenue point to a growing platform node, increasing throughput and profitability over time.
International potential: Leveraging prior infrastructure for global expansion
Delcath’s international market represents a second-layer growth engine that, while not yet monetized, has the potential to match or even exceed U.S. revenue over the next decade. Global expansion is supported by three converging factors: (1) higher liver cancer incidence outside the U.S., (2) pre-existing procedural awareness from CHEMOSAT deployments in Europe, and (3) increasingly harmonized regulatory and reimbursement frameworks that favor interventional oncology innovation.
Geographically, Asia-Pacific holds the largest concentration of unmet need. Primary liver cancers, particularly hepatocellular carcinoma (HCC), are endemic in countries like China, South Korea, Vietnam, and Thailand, where hepatitis B and C infections, aflatoxin exposure, and cirrhosis drive aggressive disease. China alone sees over 400,000 new cases of HCC annually, accounting for more than half of global incidence.
Although systemic therapies like TKIs and anti-PD-L1 combinations are standard, there remains a significant subgroup of patients who experience early progression or are contraindicated for systemic treatment, especially in older or comorbid populations. This group is well suited for liver-directed therapies like HEPZATO, particularly in the second or third line.
In Europe, Delcath has a historical footprint through its CE-marked CHEMOSAT system, which was used clinically in countries such as Germany, the Netherlands, and the UK. While that iteration lacked the integrated drug component required for reimbursement traction, it did establish procedural knowledge and hospital relationships. Some centers performed over 100 CHEMOSAT procedures, suggesting latent demand. The shift to the HEPZATO KIT, now a fully integrated drug-device combination, allows Delcath to revisit these relationships with a clearer reimbursement strategy and stronger clinical data from FOCUS.
From a regulatory perspective, Europe is moving toward more centralized device-drug combination assessments under the EU Medical Device Regulation (MDR) and EMA coordination. Because the core components of HEPZATO already have regulatory precedent and technical files on record, the path to re-certification could be materially de-risked. In parallel, national HTA bodies (e.g. NICE, IQWiG, HAS) are increasingly familiar with interventional oncology as a field, especially given growing adoption of TACE, SIRT, and ablative modalities.
Commercially, the international rollout will likely follow a phased approach: high-volume European centers (Germany, Benelux, UK) first, followed by targeted Asian markets through regional partnerships. Importantly, Delcath may leverage distributors, JV structures, or territory-specific licensing models to reduce upfront capital needs. While timelines remain to be disclosed, the commercial architecture, repeat use, site-based scaling, procedural entrenchment, will function similarly to the U.S., making international revenue both high-margin and strategically reinforcing.
The strategic implication: Platform economics that compound with indications
Delcath’s market opportunity is not just large, it is architecturally compounding. Each new indication expands TAM, but more importantly, it increases revenue per site, per patient, and per year. Because the underlying platform remains constant, and because hospital systems are already investing in onboarding, every additional use case adds leverage to the same infrastructure. This is the hallmark of a true procedural platform, akin to Intuitive Surgical’s da Vinci system or Varian’s radiotherapy suites, not a traditional biotech pipeline with siloed molecules.
This is critical for long-term investors. It means Delcath doesn’t have to rebuild from scratch with every label expansion. Regulatory approvals, reimbursement negotiations, and sales force investments made today are reused across future indications, dramatically improving capital efficiency and reducing commercial friction. Furthermore, once the platform is standard-of-care for one tumor type (e.g., CRLM), adoption for adjacent indications becomes exponentially easier due to institutional familiarity and payor precedent.
In a capital-constrained biotech environment, Delcath offers a rare thesis: one where clinical development maps directly to durable commercial returns. Every dollar spent on trials builds not just a new revenue stream but enhances the value of all existing ones. As each layer is added, first mUM, then CRLM, then iCCA and beyond, the commercial flywheel accelerates. Delcath is not just expanding into new indications; it’s compounding a liver-directed oncology platform, built to dominate an underpenetrated, high-need therapeutic category.
Delcath’s market isn’t just growing, it’s compounding. Each new indication doesn’t just add volume; it strengthens site economics, accelerates adoption, and increases per-patient value without inflating overhead. This is the structural advantage of platform oncology: a modular system with cross-indication leverage and deep procedural entrenchment.
As more cancers are brought into the treatment scope, the entire system becomes harder to displace and more profitable to scale. For investors, that means this isn’t a single-product company, it’s a liver-focused flywheel with multi-layered operating leverage and billion-dollar potential.
Market size means little without access, and in oncology, access is defined by regulatory approval and reimbursement.
9. Regulatory & Reimbursement Dynamics
Delcath’s FDA approval wasn’t just a greenlight, it was a platform unlock, creating a clear, repeatable regulatory and reimbursement path across multiple indications and geographies.
Delcath’s commercial potential hinges not only on clinical data but on its ability to navigate the twin pillars of biotech value creation: regulatory clarity and reimbursement certainty. With FDA approval for HEPZATO in metastatic uveal melanoma, the company has crossed one of oncology’s hardest thresholds, and in doing so, validated an entire procedural architecture that now serves as a regulatory template for future expansions.
The company’s proactive approach to coding, payer alignment, and health technology assessment means it’s not just playing to win in the U.S., but building a global strategy that scales with the same economic and procedural logic.
U.S. regulatory trajectory: A hard-earned approval and platform validation
Delcath’s U.S. regulatory journey has been long, complex, and ultimately value-creating, not simply because it secured FDA approval, but because of the way in which the company earned that approval. The HEPZATO KIT was approved by the U.S. FDA in August 2023 for the treatment of metastatic uveal melanoma (mUM) with liver-dominant disease. This came after more than a decade of engagement with the agency, including a complete clinical redesign following a failed 2013 NDA submission.
That prior rejection, based largely on safety concerns and insufficient evidence of efficacy, became a critical inflection point. It forced Delcath to adopt a more rigorous approach, invest in a purpose-built pivotal trial (FOCUS), and improve every aspect of its platform, including drug delivery, filtration, procedural protocol, and endpoint validation.
The resulting FOCUS trial (NCT02678572) was not a marginal iteration, it was a foundational reset. It enrolled 91 patients across multiple high-volume U.S. centers and employed both objective response rate (ORR) and progression-free survival (PFS) as key endpoints. It was powered to demonstrate superiority to best alternative care, while addressing earlier concerns about toxicity and extrahepatic progression.
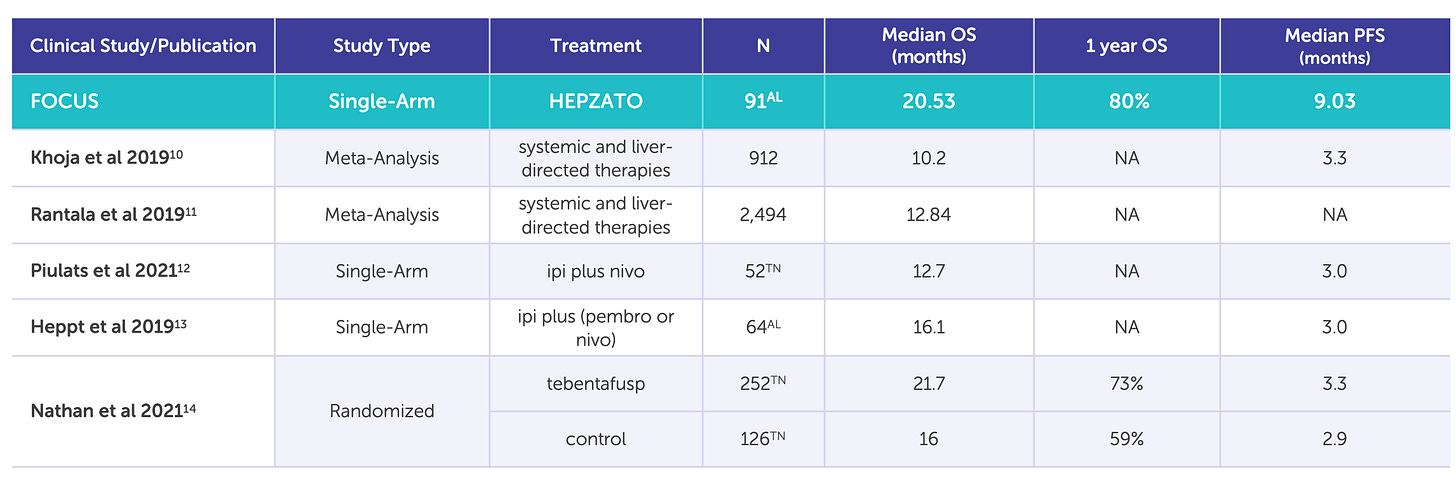
In doing so, it delivered compelling evidence that isolated hepatic perfusion with HEPZATO offers clinically meaningful benefits with an acceptable safety profile. Importantly, the trial was submitted under the 505(b)(1) regulatory pathway, the FDA’s most rigorous standard for new molecular entities or drug-device combinations. This path requires full safety and efficacy data and grants a more durable commercial foundation than 505(b)(2) bridge routes or Humanitarian Use Device (HUD) pathways, which often result in narrower labels and more fragile reimbursement.
The strategic implication for investors is that Delcath now has a high-integrity, FDA-sanctioned platform architecture. Every future indication, from CRLM to iCCA or HCC, can be pursued through supplemental NDAs (sNDAs) rather than full-blown new submissions. This substantially lowers development risk, cost, and time-to-approval for new tumor types. Moreover, the agency’s acceptance of HEPZATO’s delivery mechanism and filtration technology validates the core physics of the platform, making future iterations (e.g., automated perfusion, enhanced drug payloads, or real-time monitoring) more likely to be viewed as label expansions rather than re-evaluated devices.
In a sector where many platform stories are built on hopeful extrapolation, Delcath’s regulatory strategy is now grounded in formal FDA precedent, not theoretical modularity. This is a major advantage in both investor risk assessment and commercial scalability.
U.S. reimbursement structure: Coding, payment, and economic alignment
With FDA approval secured, Delcath’s near-term execution risk is increasingly tied to reimbursement, specifically, its ability to ensure that hospitals, physicians, and payers are economically aligned to adopt and sustain the HEPZATO procedure.
Because this is a drug-device combination administered via a complex interventional radiology (IR)-led protocol, reimbursement must cover not only the cost of the drug, but also physician services, facility use, anesthesia, filtration hardware, and peri-procedural care. Delcath’s ability to harmonize these layers determines how rapidly hospitals can onboard and how profitable each procedure is to the institution.
The company has already taken key steps toward reimbursement clarity. On the facility side, the procedure currently maps to MS-DRG 039, a Medicare Severity Diagnosis-Related Group that covers extracorporeal chemotherapy perfusion. This designation provides inpatient hospital reimbursement for the procedural cost and infrastructure utilization, and can be refined as utilization expands and coding improves.
On the physician side, the procedure leverages existing CPT codes for percutaneous hepatic arterial catheterization and extracorporeal filtration, allowing interventional radiologists to bill for time, skill, and resources, especially in academic centers where IR is a profit center.
The most strategically important layer, however, is drug reimbursement. Because HEPZATO is a single-use combination product, its economic value needs to be preserved within reimbursement frameworks. Delcath is pursuing separate reimbursement under Medicare Part B for the drug component of the HEPZATO KIT, likely through assignment of a J-code.
J-codes are essential for outpatient settings and physician-administered therapies because they allow providers to bill directly for the drug, separate from procedural or facility charges. Delcath has already submitted a permanent J-code application, which, if approved, will establish clear and consistent payment across all major payers.
Until a permanent J-code is assigned, the company is using a temporary C-code, which provides transitional coverage. This is standard practice and allows early commercial sites to receive drug reimbursement while CMS finalizes long-term pricing. C-codes typically convert to J-codes within one calendar year, assuming appropriate data submission, coding justification, and utilization metrics.
Reimbursement at private payers (e.g., UnitedHealth, Anthem, Aetna) is often influenced by CMS precedent but may require additional coverage decisions, medical policy alignment, or case-by-case adjudication early in launch. Delcath has stated that it is engaging with payers through its market access and health economics teams, and early commercial experience will likely determine speed of expansion. Fortunately, mUM’s orphan status, clear diagnosis code, and lack of competing therapies provide a clean reimbursement narrative.
From an investor perspective, the reimbursement structure is critical not only to near-term revenue realization but to the long-term margin profile of the business. A successful J-code assignment combined with stable DRG and CPT pathways will allow for pricing power, ASP durability, and commercial predictability, all of which support gross margin expansion as volume scales.
Future indications and regulatory pathways: Reducing time-to-label expansion
One of the most strategically valuable outcomes of Delcath’s U.S. FDA approval is not just the approval itself, but the platform it unlocks for future label expansions through supplemental new drug applications (sNDAs). Because the HEPZATO KIT was approved via the rigorous 505(b)(1) pathway, all subsequent indications using the same core platform, melphalan as the chemotherapeutic agent and the percutaneous hepatic perfusion (PHP) system as the delivery mechanism, can be pursued via sNDAs. This dramatically shortens the regulatory pathway, de-risks development timelines, and offers investors a clearer view into the expansion roadmap.
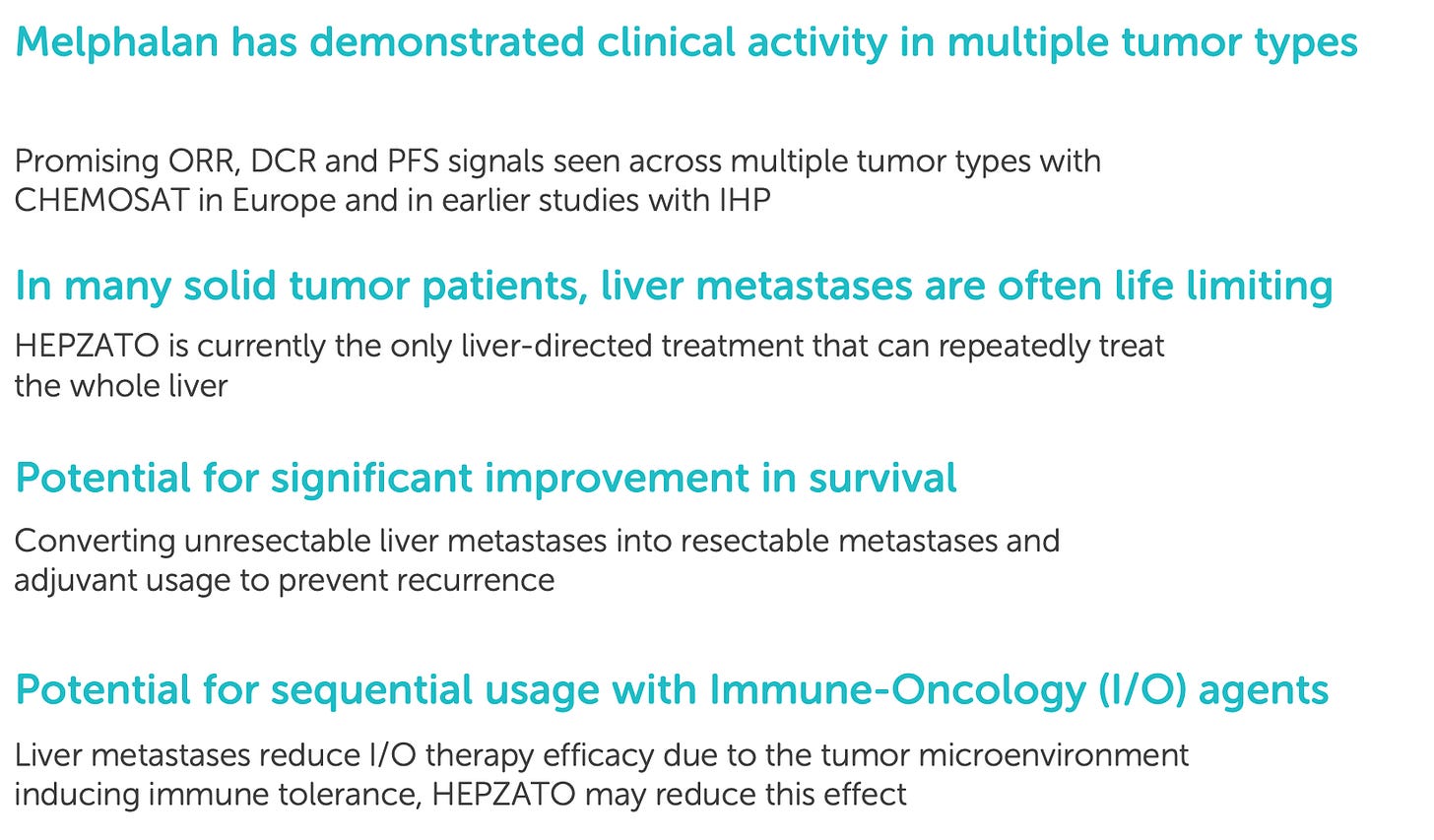
The implications for pipeline development are significant. Rather than reinventing clinical trial design or renegotiating regulatory expectations for every new tumor type, Delcath can now build on a standardized regulatory architecture. Clinical trials in colorectal cancer liver metastases (CRLM), intrahepatic cholangiocarcinoma (iCCA), or hepatocellular carcinoma (HCC) can be tailored in size and structure without the burden of duplicating platform safety studies. This flexibility allows for smaller, indication-specific trials that focus on efficacy and hepatic progression endpoints while leveraging the already-established safety and pharmacokinetic profiles.
Moreover, Delcath’s expanding clinical evidence will increasingly support the argument that the HEPZATO platform is not merely a drug-device pairing, but a modular liver-targeting platform. Each additional indication not only opens new TAM but strengthens the overall regulatory case for the procedure as a liver-specific modality, which can be extrapolated to other hepatic-dominant tumors. Over time, this may allow the company to pursue tumor-agnostic labels focused on liver-confined progression, similar to what companies like Loxo or Foundation Medicine have achieved in histology-agnostic approvals, though anatomically rather than genetically defined.
The regulatory environment is also trending in Delcath’s favor. FDA divisions within the Oncology Center of Excellence (OCE) have increasingly emphasized organ-specific burden of disease and are showing openness to alternative endpoints, such as real-world evidence (RWE), patient-reported outcomes (PROs), and progression-free survival in niche populations. Given that many of Delcath’s target patients are in the third-line+ setting with poor systemic options, the threshold for demonstrating clinical value is favorable.
Taken together, the company is positioned to move faster, with lower capital intensity, toward multi-indication commercial use, a rare structural advantage in oncology, where most companies start from zero with every label.
European regulatory landscape: From legacy CE mark to modern commercial strategy
Delcath's presence in Europe predates its U.S. approval, and while its earlier efforts under the CHEMOSAT brand faced structural reimbursement challenges, they laid the groundwork for future international expansion. Between 2012 and 2018, the company placed PHP systems in over a dozen European centers under CE mark clearance.
Though this iteration lacked an integrated drug component and was thus commercially limited, the procedural foundation it created remains relevant. These hospitals gained experience with hepatic perfusion techniques, and some performed dozens or even hundreds of cases, establishing interventional radiology teams and workflow familiarity that can now be reactivated under the updated HEPZATO KIT.
The current European regulatory framework has evolved substantially under the new Medical Device Regulation (MDR), which went into effect in 2021. For drug-device combinations like HEPZATO, MDR requires not only CE marking of the device but also review by a notified body in consultation with the European Medicines Agency (EMA).
While this adds complexity, Delcath is entering the process with key advantages: a U.S.-approved product, validated manufacturing systems, and existing procedural data from European experience. Furthermore, the EMA has shown increasing flexibility in evaluating therapies for high-unmet-need cancers, particularly when standard of care is poor and the therapeutic modality is already well understood.
Health technology assessments (HTAs) represent the next regulatory step post-approval and are essential to unlock reimbursement across individual countries. Delcath is expected to pursue early engagement with HTA bodies in core markets like Germany (IQWiG/G-BA), the UK (NICE), France (HAS), and the Netherlands (ZiN).
mUM represents a favorable entry point due to its rarity, lack of effective systemic therapies, and the presence of centralized treatment networks, such as ocular melanoma referral centers. In Germany, where Delcath previously operated under a clinical reimbursement framework, early re-activation of prior sites could expedite commercial adoption with minimal new investment.
The opportunity in Europe is not limited to mUM. Many of the company’s expansion indications (CRLM, iCCA, HCC) have higher per-capita incidence rates in Europe than in the U.S., driven in part by population age structure and delayed screening.
Additionally, European oncologic practice often favors regional therapies earlier in the disease course, making PHP potentially more relevant in second-line settings compared to the U.S., where systemic therapy dominates. Delcath will likely tailor its go-to-market strategy on a country-by-country basis, depending on procedural volume, HTA timelines, and reimbursement complexity.
This may include direct sales in priority markets like Germany and the UK, and distributor models in smaller or slower-moving jurisdictions. The overarching takeaway for investors is that Europe is not a speculative frontier, it is a re-entry market with existing procedural precedent, regulatory engagement, and potential for rapid incremental revenue at relatively low marginal cost.
Global harmonization and strategic leverage
As the global oncology landscape continues to evolve, Delcath is strategically positioned to benefit from converging regulatory, procedural, and reimbursement trends across major geographies.
Interventional oncology, once viewed as a niche or experimental discipline, is now increasingly recognized as a core therapeutic pillar, alongside medical oncology, radiation oncology, and surgical resection. This shift is not just academic. It reflects fundamental changes in how cancer is treated, reimbursed, and regulated across diverse health systems.
One of the most significant trends supporting Delcath’s global positioning is the harmonization of regulatory frameworks for drug-device combination products. The FDA, EMA, Japan's PMDA, and Health Canada have all adopted more integrated review structures for such platforms, acknowledging that therapeutic innovation increasingly spans beyond traditional chemical entities.
As agencies adapt, companies like Delcath with validated platform architectures and high unmet-need targets are better able to synchronize approval strategies, streamline submissions, and avoid redundant regulatory costs.
In parallel, the standardization of procedural coding and reimbursement language, such as CPT codes, Diagnosis-Related Groups (DRGs), and HCPCS (J-codes) in the U.S., or OPCS and EBM in Europe, allows for greater transferability of economic models.
This means that the arguments used to secure U.S. reimbursement (e.g., cost-offset vs systemic therapy, hospital margin potential, procedural precedent) can increasingly be repurposed in international payer negotiations. As clinical outcomes data matures across indications, Delcath can lean on a centralized pharmacoeconomic narrative that translates across HTA bodies and commercial insurers.
Emerging markets offer an additional layer of optionality. While countries like China, Brazil, India, and parts of Southeast Asia are often challenging from a regulatory standpoint, they also account for a disproportionately large share of global liver cancer incidence, especially HCC.
These markets may not be suitable for direct commercialization in the near term, but they present viable opportunities for regional licensing, distribution partnerships, or joint ventures. A capital-light approach in these geographies could allow Delcath to access high-volume patient populations with minimal overhead, while retaining upside through royalties or milestone-based structures.
Importantly, as Delcath builds clinical, procedural, and economic credibility in the U.S. and Europe, its ability to negotiate on favorable terms in international markets strengthens. Global harmonization isn't just about regulation, it's about leverage.
The more Delcath succeeds in one system, the more transferable its model becomes across others. Over the long term, this global replicability acts as a force multiplier, enhancing the platform’s margin profile, accelerating adoption curves, and reinforcing competitive moat through entrenched hospital infrastructure.
Delcath has quietly constructed one of the most elegant platform regulatory strategies in oncology: win once, scale many. By securing FDA approval under the rigorous 505(b)(1) pathway, and layering reimbursement through a blend of existing CPT, DRG, and (soon) J-code structures, the company has de-risked the most complex parts of oncology commercialization.
Add to that a European regulatory relaunch, international procedural familiarity, and increasing global harmonization for combination products, and what emerges is not a single-market bet, but a globally defensible, multi-indication system with the infrastructure to scale intelligently and the regulatory traction to do it efficiently.
With the path to market established, we can now evaluate how Delcath stands apart from competitors, or doesn’t.
10. Competitive Positioning & Differentiation
Delcath’s edge lies not in a single drug or device, but in a procedurally entrenched oncology platform that becomes stronger, stickier, and more defensible with every patient treated and every hospital activated.
In oncology, real competitive advantage rarely comes from molecule alone, it comes from integration. Delcath’s platform is not a theoretical modality, but a deeply embedded procedural ecosystem with clinical, regulatory, and institutional lock-in. By targeting liver-dominant cancers through a high-dose, regionally delivered chemoperfusion procedure, Delcath has positioned itself in a category of one: not competing with systemic therapies, but complementing and outlasting them via infrastructure-scale defensibility. Its moat is not molecular exclusivity, it’s operational complexity, institutional inertia, and category authorship.
Procedural moat: Defensibility through institutional inertia and clinical complexity
Delcath’s most important and misunderstood competitive advantage lies in the procedural nature of its therapy. While many investors initially perceive this complexity as a commercial liability, compared to traditional infused or oral drugs, it functions as a powerful moat. The HEPZATO KIT is not a standalone pharmaceutical; it is a regulated, multi-step, high-skill procedure that requires a hospital’s coordinated deployment of interventional radiology, anesthesiology, oncology, and intensive monitoring.
To administer it safely and effectively, a center must commit clinical time, technical training, procedural capacity, and financial resources. Once implemented, this system becomes institutionally embedded, creating a formidable layer of inertia that few new entrants can displace.
At the center level, onboarding HEPZATO is a non-trivial investment. Hospitals must credential physicians, reallocate OR or IR lab time, train multidisciplinary teams, establish inventory protocols for a just-in-time kit with a short shelf life, and coordinate with internal reimbursement departments. This onboarding process can take 4-6 months, but once completed, it transforms the center into a revenue-generating node for the company with increasing marginal returns per use.
Each subsequent procedure becomes easier, faster, and more efficient, creating a site-specific economy of scale. Staff familiarity improves patient throughput, IR teams optimize perfusion time, and billing departments streamline reimbursement. In short: every additional procedure increases the institution’s internal ROI, and deepens their commitment to the platform.
This is where Delcath’s advantage compounds. Unlike pharmaceutical products, where competition can be introduced via price undercutting, new formulations, or label creep, the infrastructure lock-in of a procedural oncology platform makes HEPZATO a category of one. Any future competitor would need to replicate not just clinical efficacy, but also operational compatibility with existing hospital workflows.
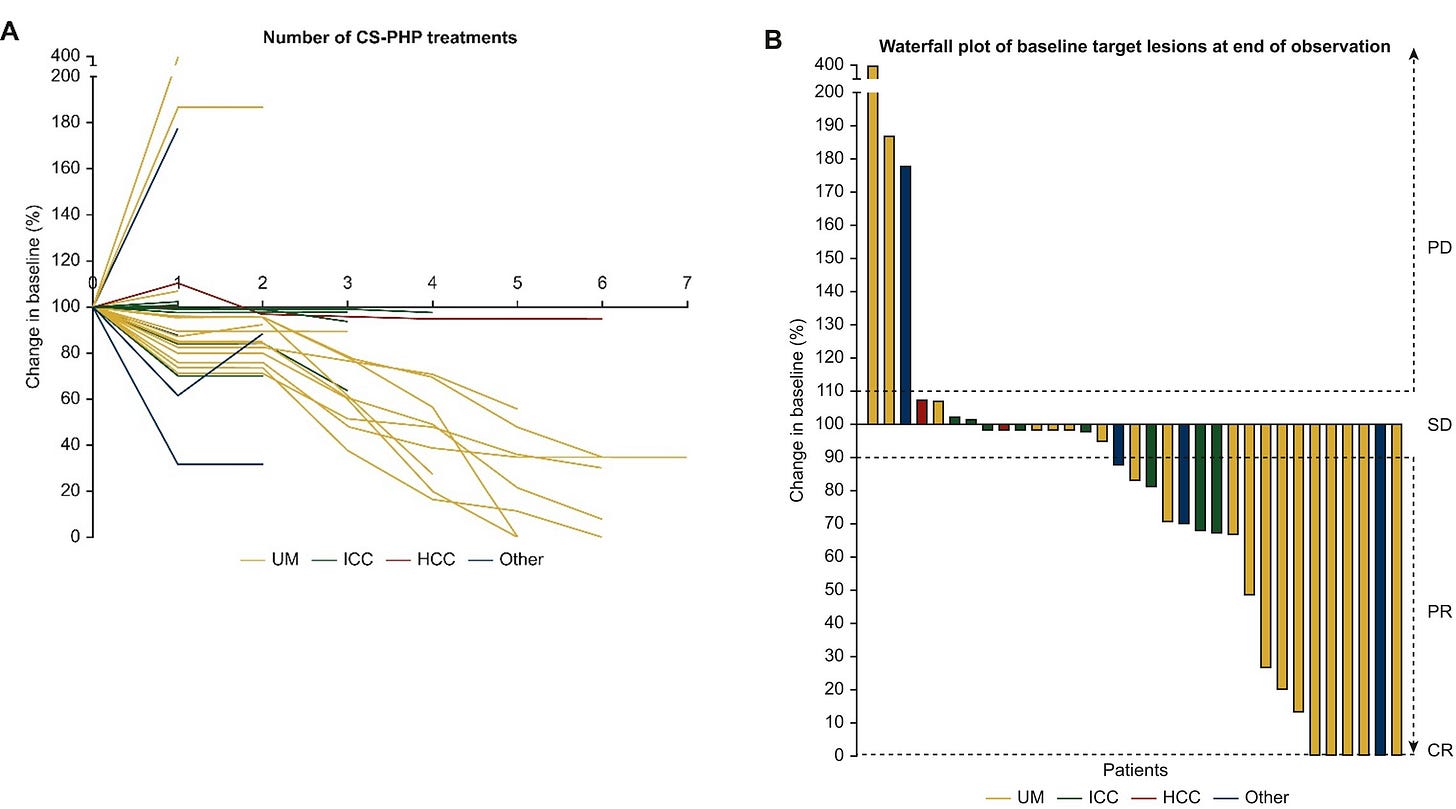
Even in the rare case where a rival develops a marginally superior drug or perfusion technology, the switching cost, both real and perceived,for the hospital is enormous. Re-credentialing, retraining, reimbursement renegotiation, and physician re-education act as deterrents to disruption. In this context, HEPZATO becomes not just a product, but an institutional habit.
Delcath is already taking advantage of this dynamic through strategic placement. The company is deliberately targeting large, high-volume academic medical centers, those with multiple IR labs, cross-specialty coordination, and trial participation capacity. These institutions also tend to treat patients across multiple tumor types, meaning that once trained on HEPZATO for mUM, they can quickly expand into treating CRLM, HCC, or iCCA with no added procedural training.
This enables a form of stacked monetization: multiple indications flowing through a fixed infrastructure base. Once 30-50 sites are operational, the economics of adding a new indication become highly favorable, both for the company and the institution.
In the longer term, this procedural moat also creates negotiation leverage with payers and partners. Hospitals that are procedural incumbents tend to defend their capabilities, securing formulary status, advocating for coverage, and resisting system-wide transitions to unproven alternatives. Delcath is not trying to win prescribing behavior at the physician level. It is capturing structural commitment at the institutional level, a stickier, more defensible layer of commercial traction. This model, while slower to initiate, becomes exponentially more efficient once momentum builds.
In this regard, HEPZATO resembles not a drug, but a platformed procedural franchise, akin to Intuitive Surgical’s da Vinci, Edwards Lifesciences’ TAVR system, or Boston Scientific’s Watchman. These systems created entirely new procedure classes, trained centers over time, and built lasting economic ecosystems around their use. Delcath’s opportunity is similar: to embed a new standard of hepatic perfusion into the infrastructure of tertiary cancer care, one site at a time, with compounding effect.
Orphan indications as a strategic beachhead: The platform value of mUM
While metastatic uveal melanoma (mUM) affects a small number of patients,estimated at ~1,200 annually in the U.S., its strategic importance to Delcath lies not in its revenue potential alone, but in its catalytic role within the company’s broader platform strategy.
Rather than treating mUM as a standalone commercial product, Delcath has intelligently used it as a strategic beachhead: a narrow, under-served, yet clinically urgent entry point to build institutional trust, procedural adoption, and regulatory precedent. In this context, mUM is not the core value driver of the business, it is the activation mechanism for everything else that follows.
The disease itself is ideal for a first-in-market hepatic perfusion therapy. Over 90% of metastatic uveal melanoma cases involve liver-dominant disease, and systemic options for this population are largely ineffective. The historical standard of care includes chemotherapy, checkpoint inhibitors, or clinical trial enrollment, all of which deliver poor response rates.
Tebentafusp (KIMMTRAK), while recently approved, is only effective in HLA-A*02:01, positive patients and has limited activity in visceral metastases, particularly in the liver. For patients with liver-only or liver-predominant progression, mUM is a clearly defined, high-need anatomical problem that systemic agents are ill-equipped to solve. This makes it an ideal target for a high-dose, liver-isolated intervention like HEPZATO.
From a regulatory perspective, mUM offered a manageable pathway to approval. Its orphan disease status qualified Delcath for multiple FDA support mechanisms, including reduced review timelines, guidance access, and post-marketing flexibility. It also allowed for a smaller, single-arm trial (FOCUS) with well-defined hepatic response endpoints, acceptable to the FDA given the clear unmet need and lack of existing options.
Importantly, this trial and approval now create a precedent for future supplemental NDAs. By proving safety and efficacy in this rare indication, Delcath has demonstrated that its procedural platform meets the FDA’s evidentiary burden for liver-directed therapy, a critical validation for expansion into more competitive, higher-volume cancers.
Equally important is the institutional impact of launching through mUM. Because of the disease’s rarity and severity, patients are typically referred to a handful of high-acuity academic medical centers, many of which also serve as regional hubs for other liver metastasis populations. These are exactly the institutions Delcath must activate to scale its platform.
By introducing HEPZATO into these sites through the lens of a rare disease, the company reduces internal resistance. Physicians are more willing to trial novel therapies in orphan settings, hospital administrators are more likely to approve use for compassionate or urgent need, and payer pushback is lower when no alternative options exist. In short, mUM provides permissionless entry into institutions that are otherwise difficult to penetrate.
Once trained and credentialed, these sites form the core of Delcath’s “scalable spine” for expansion. The procedural knowledge, inventory systems, billing codes, and care pathways built for mUM are immediately reusable when the company gains additional indications like CRLM, HCC, or iCCA.
This provides a major platform operating leverage: Delcath will not need to retrain staff or re-negotiate institutional protocols. Instead, it can simply expand eligible patients through updated labels, monetizing new clinical verticals without incurring onboarding costs again.
Furthermore, early success in mUM creates a positive feedback loop across stakeholders. Clinicians develop comfort and confidence with the therapy, which accelerates enrollment in future trials. Early adopters become key opinion leaders, presenting data at conferences and advocating for expanded access.
Payers begin to establish precedents for coverage, smoothing reimbursement negotiations for new tumor types. Regulators see real-world safety outcomes that support label expansion or procedural flexibility. This is how rare diseases, when chosen wisely, can unlock disproportionate strategic value in oncology platform companies.
In this light, mUM is less a commercial product than a strategic wedge. It offers Delcath a rare opportunity to control its own commercial narrative: entering institutions through a disease that is both tragic and underserved, proving procedural viability under the least contested conditions, and then scaling horizontally across liver-dominant tumors. It is the first domino in a much larger cascade, and its value should be measured not by peak revenue, but by its role in seeding Delcath’s entire delivery network.
Market positioning: A category of one in liver-dominant oncology
Delcath occupies a strategic position in oncology that is rarely available to emerging companies: it operates in a high-burden, clinically underpenetrated anatomical niche where standard-of-care options are limited, and competition is fragmented or misaligned. While most oncology companies define themselves around molecular targets, Delcath’s approach is organ-specific, anchored not to histology but to geography.
The liver, as a site of metastatic burden, is both a convergence point for systemic cancers (e.g. colorectal, breast, pancreatic) and the origin of several primary malignancies (e.g. HCC, iCCA). In treating liver-dominant tumors, Delcath is not developing yet another systemic therapy, it is delivering high-dose regional chemotherapy to a discrete, functionally vital organ in a way that systemic agents fundamentally cannot.
This positioning matters strategically because it creates category asymmetry. Delcath is not competing directly with PD-1 inhibitors, small molecule TKIs, or ADCs. It is not vying for formulary space against orally dosed, broadly active agents. Instead, it occupies a therapeutic trench that systemic therapies consistently fail to reach: patients with isolated or liver-predominant progression, often following prior lines of systemic treatment.
These patients are ideal candidates for liver-directed therapy, especially when hepatic disease is driving prognosis, liver function is preserved, and other options have been exhausted. Yet the market has historically lacked an effective, standardized approach to this anatomical problem.
Previous interventions in this space, such as Yttrium-90 radioembolization (TheraSphere, SIR-Spheres) or transarterial chemoembolization (TACE), offer only partial solutions. They rely on embolic particles or radioactive isotopes delivered intra-arterially but lack the filtration and drug concentration controls of Delcath’s PHP system. They also have limited applicability outside of HCC and are generally not used in metastatic settings like colorectal cancer.
More importantly, they are not paired with a systemic chemotherapy platform that delivers high, liver-concentrated doses of an established cytotoxic like melphalan. This gives Delcath a unique pharmacokinetic signature: it can administer drug levels that would be systemically lethal, but isolate their effects to the liver, achieving both efficacy and tolerability.
Delcath’s ability to bridge multiple histologies through a shared organ focus gives it platform leverage. Rather than building independent development programs for each tumor type, it can extend use across indications as long as the liver is the dominant site of progression. This creates operational and regulatory efficiencies uncommon in oncology.
For example, the same interventional radiologist at Memorial Sloan Kettering could use HEPZATO on an mUM patient, a CRLM patient, and an HCC patient, all with the same equipment, perfusion technique, and institutional pathway. Each new tumor label expands the horizontal scope of the platform while deepening procedural utilization at each site.
From a commercial perspective, this positioning also allows Delcath to avoid direct competition with large pharma. The company is not trying to displace Merck’s Keytruda or Roche’s Tecentriq in first-line treatment algorithms. Instead, it complements systemic agents by offering salvage or consolidation therapy in patients whose disease becomes localized to the liver. In the case of CRLM, this could mean treating a patient who has already progressed on FOLFOX and anti-EGFR therapy.
In HCC, it could mean delivering high-dose therapy to non-embolization candidates who failed TKIs or immunotherapy. This “adjacent positioning” makes Delcath partnerable rather than threatening, enabling future collaborations with companies who want to extend their systemic agents’ durability or downstream utility.
Equally important, the company’s anatomical approach plays directly into emerging trends in oncology trial design and regulatory science. As the FDA continues to support tissue-agnostic or site-specific approvals based on biomarker or anatomic criteria (e.g., tumor mutational burden, MSI-H, NTRK fusions), the precedent for non-histology-based drug platforms becomes stronger.
Delcath is anatomically agnostic in terms of tumor origin, but anatomically precise in its delivery. It fits neatly into a world where regulators are moving away from rigid histology boundaries and toward functional definitions of therapeutic value.
The net result is a company that exists in a category of one, not competing against first-line systemic drugs, not interchangeable with embolic radiotherapies, and not easily replicated by oral or infused agents. It is this differentiation, anchored in the physical constraints of drug delivery, the anatomical logic of metastasis, and the infrastructure inertia of procedural oncology, that gives Delcath its edge. The company is not trying to win a crowded race. It is building its own lane, defined by the liver’s role in metastatic progression and the need for precision, high-dose, regional therapy.
Data & IP as long-term assets: Building a durable advantage through clinical experience and regulatory positioning
While Delcath is not a traditional biotech company built on novel molecular patents, it is nonetheless assembling a differentiated and defensible intellectual property and data position that strengthens over time. Its moat is not built primarily on composition-of-matter claims, but rather on a cumulative body of procedural data, regulatory approvals, device-specific know-how, and real-world clinical experience that is difficult to replicate and increasingly valuable as the platform scales.
First, the company owns and operates under a suite of patents and regulatory protections related to the PHP system, filtration technology, and procedural configuration of the HEPZATO KIT. These patents cover not only aspects of the perfusion system and circuit, but also drug-device integration, safety features, and filter design, extending through 2033 and beyond in various jurisdictions.
While these do not provide monopoly protection in the same way a small-molecule composition might, they are meaningful in deterring exact replication of the platform. More importantly, because the product is regulated as a drug-device combination under FDA’s 505(b)(1) pathway, any follow-on entrant would need to conduct extensive clinical trials to demonstrate both efficacy and safety for a new delivery mechanism, a high-risk, capital-intensive undertaking.
The more powerful moat, however, lies in procedural data and accumulated real-world experience. With each additional center trained, patient treated, and procedure reimbursed, Delcath is generating institutional knowledge and operational precedence that cannot be shortcut by competitors.
These are not abstract “data assets” in the software sense, they are embedded workflows, refined protocols, interventional radiologist expertise, and payer coding relationships that make the HEPZATO procedure easier and more defensible with each use. Just as Intuitive Surgical benefits from the accumulated procedural fluency of thousands of surgeons trained on da Vinci, Delcath will benefit from its procedural incumbency, a feedback loop in which data and practice reinforce each other.
On the regulatory side, the successful approval of HEPZATO under the full NDA process gives Delcath an intangible but substantial head start. It has navigated the FDA’s Oncology Center of Excellence, submitted validated manufacturing protocols, and received buy-in on trial design, endpoints, and safety profiles.
Future sNDAs for additional indications can leverage the existing regulatory framework, reducing risk and cost. Any competitor would have to start from scratch, conducting a full clinical development program without the benefit of a procedural precedent. Moreover, the fact that HEPZATO is already integrated into CMS billing codes, hospital formularies, and academic center workflows adds non-technical friction to market entry for any fast followers.
Delcath is also well-positioned to build a proprietary dataset around hepatic perfusion that no competitor can access. As its registry grows and multi-indication trials proceed, the company will accumulate prospective and retrospective data on patient selection, response rates, durability, adverse events, and long-term outcomes across multiple tumor types.
This creates a proprietary knowledge base that informs clinical guidelines, payer dossiers, and KOL adoption. Over time, the company could even standardize its own real-world evidence framework, a valuable asset as payers and regulators increasingly incorporate observational data into their assessments of therapeutic value.
Finally, there is a first-mover advantage in shaping clinical and commercial standards. As the only company with an approved, perfusion-based chemotherapy system regulated as a drug-device combo in the U.S., Delcath is now writing the rules for its category. It will define what trial endpoints are acceptable, how adverse events are coded and mitigated, what procedure duration is typical, and how cost-benefit analyses are framed.
In oncology, where precedent carries substantial weight, this influence becomes a long-term asset. It positions Delcath not only as a product provider, but as a category architect, a company whose experience and expertise become central to how hepatic perfusion is understood and reimbursed globally.
While Delcath does not own a novel molecule in the traditional biotech sense, its asset base is nonetheless deep, multi-dimensional, and strategically structured for durability. Through a combination of IP, procedural data, regulatory experience, and payer integration, the company is building a high-friction environment for potential competitors, one that grows stronger with each treated patient and activated center. For long-term investors, this makes the business increasingly self-reinforcing and incrementally more defensible with time.
Organizational focus and execution discipline: A culture built for operational precision, not narrative hype
While many early-stage biotech and medtech companies burn capital chasing platform potential without clear operational pathways, Delcath’s emerging competitive advantage also lies in its internal discipline. The company is building a business that is structurally aligned with the demands of a complex, procedural, hospital-driven oncology platform, where execution, cross-functional coordination, and institutional adoption matter more than flashy science or short-term milestones. This cultural and organizational focus is itself a differentiator, especially in a market where hype cycles often outrun operational readiness.
Under the leadership of CEO Gerard Michel, Delcath has deliberately shifted its identity from an R&D-heavy, capital-intensive clinical-stage biotech to a commercially focused, precision-execution platform company. This transition is evident across every layer of the organization, from its hiring strategy to its capital deployment, site activation pacing, trial design, and reimbursement execution.
Unlike biotech peers that spend heavily on multiple early-stage programs or exploratory modalities, Delcath has focused tightly on commercial readiness, streamlining its pipeline to indications with clear hepatic progression logic, and optimizing its go-to-market around high-throughput, academically anchored centers.
This cultural shift matters because the nature of the HEPZATO platform is inherently operational. It is not a pill that can be marketed via reps to community oncologists. It is a procedure that must be integrated into hospital systems, executed by cross-functional clinical teams, and justified through complex reimbursement pathways.
Success in this environment requires a company with deep cross-domain expertise: in regulatory negotiation, interventional radiology education, health economics, device logistics, and revenue cycle management. Delcath’s management has hired accordingly, bringing in operationally experienced leaders like Martha Rook (COO, ex-insitro/Sigilon) and Sandra Pennell (CFO, ex-Vericel), whose backgrounds emphasize execution over theoretical platform building.
Moreover, the company has avoided the common trap of overextending into tangential modalities or therapeutic areas. While many small-cap biotechs dilute focus by chasing immuno-oncology combinations or exploring preclinical verticals unrelated to their lead programs, Delcath has remained focused on its core: building a scalable, revenue-generating, liver-directed oncology platform.
Even its clinical trials are designed with commercial scaling in mind-targeting indications that flow through the same procedural infrastructure and can be treated by the same interventional teams. This level of internal strategic coherence is rare in small-cap oncology and is especially valuable in platform businesses that rely on deep institutional integration.
Executional discipline is also evident in how Delcath has approached reimbursement. Rather than assuming payer support will follow approval, the company engaged in early, methodical payer education and coverage groundwork. It worked preemptively with CMS, engaged MACs regionally, and structured its coding and billing strategy to match procedural reimbursement norms, reducing the lag between approval and revenue conversion.
This kind of work is invisible to the market but critical to commercial survival in the procedure-based oncology segment. For investors, it signals a company that understands not just how to develop a therapy, but how to get it paid for, delivered, and scaled in real-world settings.
Delcath has demonstrated financial discipline, maintaining a relatively modest cash burn and deliberately staging capital raises around value inflection points. This is in stark contrast to biotech peers that dilute aggressively without advancing commercial readiness. While this conservative approach may limit speculative momentum in the short term, it increases the likelihood of long-term value capture. It also enables the company to scale profitably as new indications are added, expanding utilization through existing centers, rather than requiring massive salesforce buildouts or rep-level promotion.
Delcath’s organizational advantage is not flashy. It is not based on PR, MOUs, or high-profile collaborations. It is grounded in execution: the slow, deliberate work of building a new procedural standard in oncology. In an industry where many companies confuse platform potential with business readiness, Delcath is building the rarest asset of all, a company with both.
Delcath is doing what few small-cap oncology companies ever achieve: converting technical validation into structural entrenchment. Through a combination of procedural integration, regulatory credibility, IP reinforcement, and focused execution, the company is creating a platform that becomes more defensible as it grows.
Every center trained, every indication added, and every payer aligned compounds its moat, not just against competitors, but against irrelevance. Delcath isn’t chasing hype; it’s engineering permanence. And in the long game of oncology, that is the most enduring competitive edge of all.
Strategic differentiation is necessary, but it’s not sufficient. Execution risk remains central to the thesis.
11. Clinical Risk & Executional Challenges
Delcath’s greatest challenge isn’t proving its science,it’s operationalizing a complex procedure in a fragmented system fast enough to outpace capital burn, reimbursement lag, and institutional inertia.
Despite Delcath’s differentiated platform, strong procedural moat, and disciplined leadership, the company is not without meaningful risks. These are not abstract uncertainties, they are rooted in the inherent challenges of launching a novel therapeutic modality, scaling a procedural oncology business, and navigating both regulatory and institutional complexity.
What makes Delcath investable is not the absence of risk, but rather the nature of those risks, many of which are executional and potentially mitigable, as opposed to structural or binary in nature.
Commercial execution risk: Scaling a procedural therapy across a fragmented hospital ecosystem
At this stage of its lifecycle, Delcath's core challenge is not scientific validation, it has already achieved that with the FDA approval of HEPZATO KIT for metastatic uveal melanoma (mUM). Instead, the company now enters what is arguably the most operationally demanding phase of any biotech: converting regulatory success into predictable commercial execution.
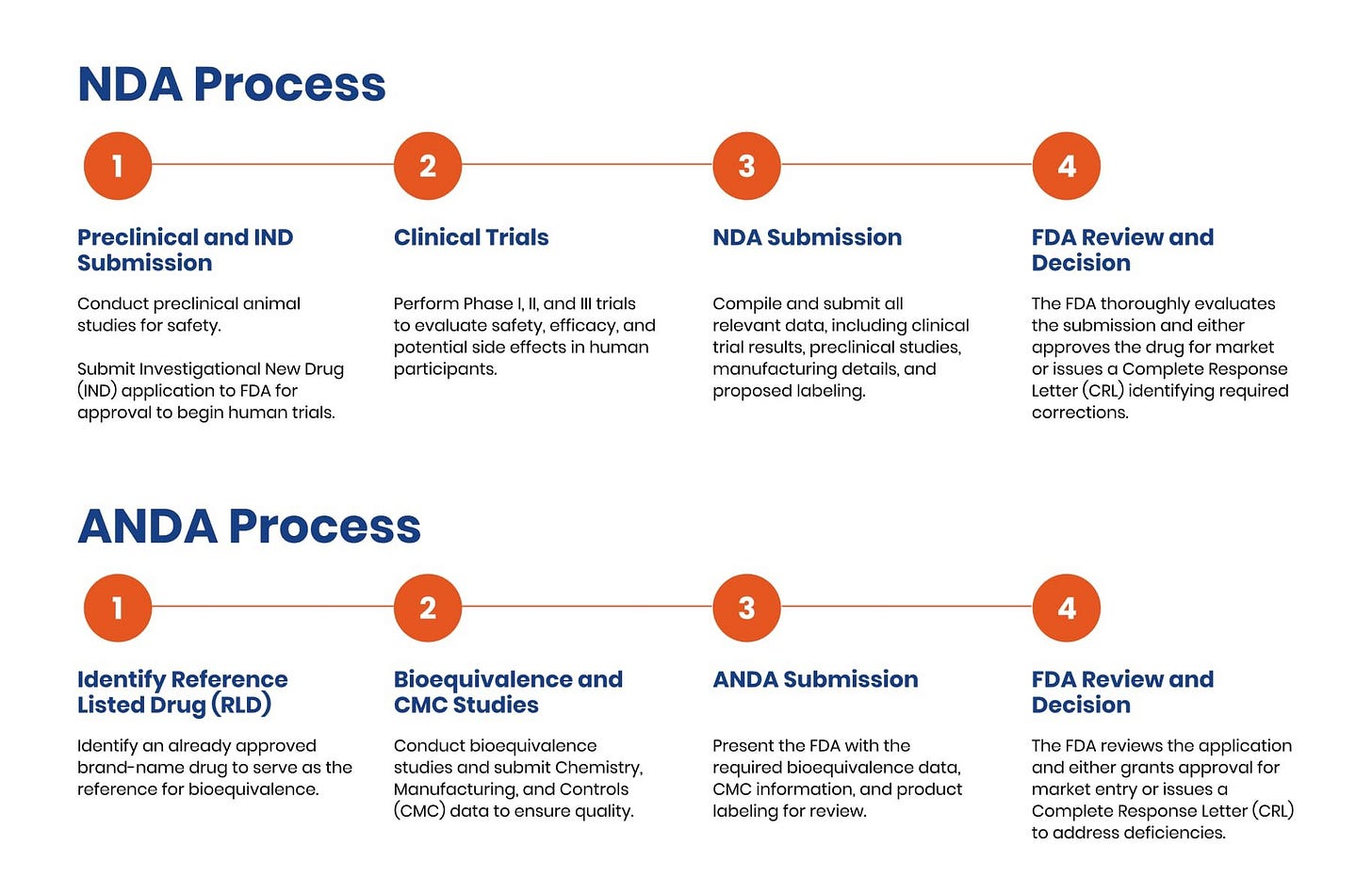
In Delcath’s case, this execution is uniquely difficult because its product is not a simple pill or infusion; it is a complex, high-touch, hospital-administered procedure. Every treatment requires coordination between interventional radiologists, anesthesiologists, oncology teams, perfusionists, inpatient beds, and the hospital billing department. This makes the sales cycle institutional, not prescriber-driven, and exposes the company to a slow, nonlinear ramp.
Unlike small-molecule drug launches, where growth can be driven by pushing out reps and securing formulary coverage, Delcath must build institutional capability one center at a time. Hospitals must internally credential physicians, train cross-functional clinical teams, schedule operating room or IR lab time, align internal inventory systems with just-in-time kit delivery, and ensure compliant billing workflows for a Category III CPT-coded procedure with limited precedent.
Even for hospitals enthusiastic about HEPZATO’s clinical value, these logistics are a major friction point. Delcath estimates a 4-6 month onboarding period per site, but this assumes no internal bureaucratic delays, staffing turnover, or scheduling conflicts. In reality, some centers could take 6-9 months to become fully operational, especially if procedural volume is low early on.
The concentration of target institutions magnifies the importance of each account. Because HEPZATO is most appropriate for patients with liver-dominant progression, treatment will be funneled into a relatively small number of academic and high-volume regional centers with the clinical infrastructure to perform it.
Delcath is not launching to a diffuse prescriber base; it is aiming to activate ~30-50 sites in the U.S. over the medium term. If 10 of those sites underperform, it could materially impact national volume and revenue realization. This means the company must be flawless in site targeting, procedural training, and support infrastructure, including field teams, clinical liaisons, and reimbursement assistance.
There is also a cultural element to commercial risk. Academic institutions are often slow to adopt new procedures, not because of skepticism, but because of internal complexity and institutional inertia. Scheduling cross-departmental training, establishing SOPs, and incorporating new technologies into multidisciplinary tumor boards takes time.
Early adoption often depends on individual physician champions, typically a lead IR or oncologist who pushes internally for site activation. Without strong clinical advocates and well-aligned administration, even promising technologies can stall in “approved but not implemented” limbo.
To its credit, Delcath appears aware of these risks and is deploying targeted mitigation strategies. The company is focusing first on early-adopting centers with mUM experience and previous trial participation, reducing initial friction. It is offering robust field support and hospital engagement resources, including site activation teams and onboarding playbooks.
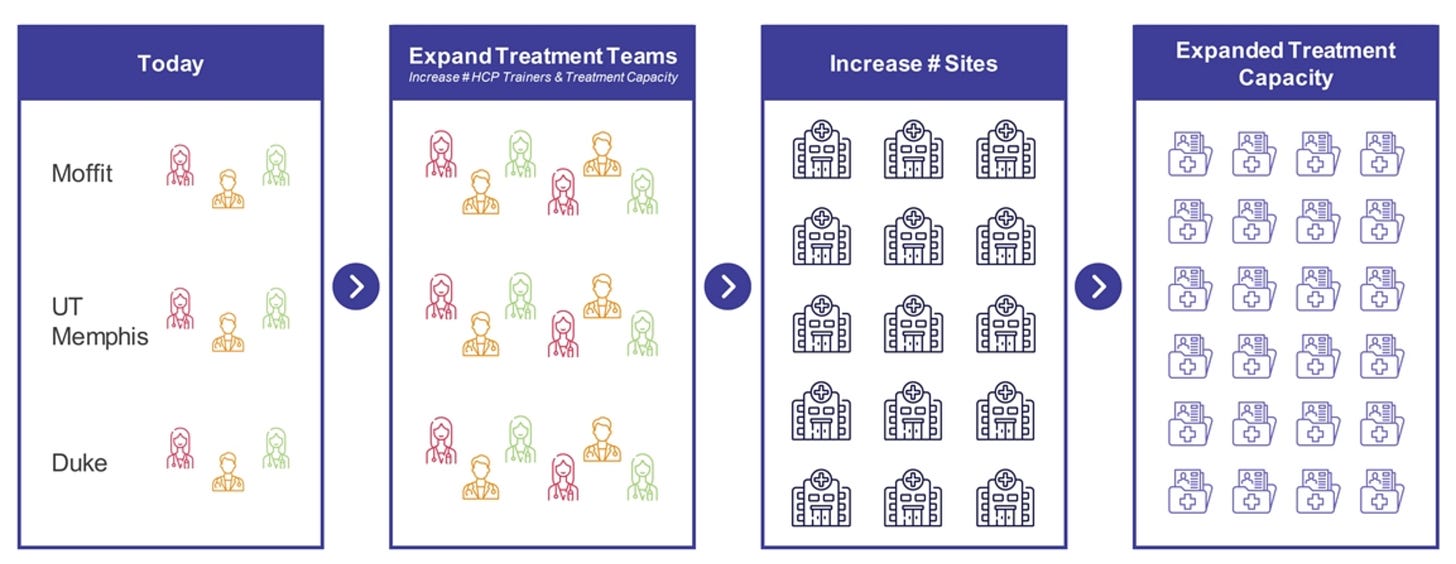
Delcath has also built an internal commercial team with deep experience in procedure-based oncology sales, rather than traditional pharma backgrounds. These are all positive signs, but they do not eliminate the underlying fragility of procedural scaling.
For long-term investors, the key question is not whether HEPZATO will be used, it will be. The question is: how quickly, how widely, and with how much institutional consistency? A procedural platform lives or dies not by scientific excitement, but by procedural inertia.
If the onboarding timeline extends, if early centers treat fewer patients than expected, or if logistical challenges overwhelm early adopters, revenue could be delayed by years. This would create a gap between platform potential and realized cash flow, forcing additional dilution or capital raises in the interim.
Commercial execution is not just a tactical hurdle for Delcath, it is the central gating factor that determines whether the company can transition from a clinically validated concept to a scalable oncology franchise. The market has seen this before: promising device-drug hybrids fail to scale not because of science, but because they couldn’t operationalize complexity at scale. Delcath must now prove that it is the exception.
Reimbursement friction: Navigating procedural complexity in a fragmented payer environment
For a platform like Delcath’s, clinical efficacy is only part of the value chain. In order for the business to scale, its procedure must also be predictably reimbursed across multiple payers and care settings. The HEPZATO KIT is a hybrid product: part high-cost chemotherapy, part procedure-dependent drug-delivery system. Its success depends not just on FDA approval, but on seamless alignment between coding, billing, and payment systems at both federal and commercial levels. Herein lies one of Delcath’s most structurally important risks: reimbursement friction.
The complexity stems from the multi-component nature of the therapy. Administering HEPZATO involves: (1) the drug kit itself, (2) the perfusion procedure, (3) physician time across multiple specialties (IR, anesthesia, oncology), (4) facility time in an OR or interventional suite, and (5) post-procedure monitoring, often requiring inpatient care.
Each of these components touches different parts of the hospital’s revenue cycle, and each must be adequately reimbursed to make the treatment financially sustainable for the institution. If even one component (e.g., facility fees or anesthesia billing) is undercompensated or ambiguous, hospitals may defer use of the therapy regardless of its clinical benefit.
Delcath has secured placement for HEPZATO under MS-DRG 039, which covers extracorporeal procedures involving chemotherapy. This creates a pathway for inpatient reimbursement under Medicare, especially for the mUM population, which skews older. However, even with a defined DRG, real-world payment can vary considerably by region, MAC (Medicare Administrative Contractor), and institutional coding practices.
Moreover, commercial payers often delay full adoption of new procedural codes, especially for combination therapies that lack precedent. This means early reimbursement may require manual adjudication, appeals, or interim authorization processes, which add administrative burden to already-complex hospital workflows.
This challenge is magnified by the procedural novelty of HEPZATO. Unlike standard-of-care infusions or oral oncology agents, it has no historical analog in payer systems. As a result, coverage policies must often be built from scratch, especially with commercial insurers.
Payers need to understand not just the clinical rationale, but also the cost-offset logic: Will patients treated with HEPZATO avoid expensive hospitalizations later? Will it delay systemic progression or expensive next-line therapies? These are not questions payers answer quickly, especially when the per-procedure cost is high and volumes are initially low.
Importantly, reimbursement delays can have disproportionate effects on procedural businesses. For a traditional drug, uncertainty in payer coverage may simply shift prescribing behavior. For a procedure like HEPZATO, uncertainty can halt institutional adoption entirely. Hospitals will not activate a procedure that risks underpayment, especially when internal champions must advocate for scarce operating room time, cross-functional training, and resource reallocation. In this way, financial ambiguity becomes clinical inertia.
Delcath has proactively addressed these challenges. The company built out a dedicated reimbursement team early in its commercial planning, began payer education campaigns pre-approval, and has provided billing guides to early centers. It is also pursuing pass-through reimbursement status and has engaged CMS in clarifying policy decisions.

These are all encouraging steps, but they don’t eliminate risk. Reimbursement is inherently slow-moving, especially for new-to-world procedures. Some MACs or commercial plans may delay or deny claims, requiring appeals and case-by-case support. This limits initial throughput and may extend the revenue curve, even as clinical demand grows.
From an investor lens, the key question is: how quickly can Delcath normalize reimbursement friction to the point where it no longer slows adoption? The danger is not outright denial, it is ambiguity. Ambiguity breeds hesitation, and hesitation in a hospital means deferred treatment. If too many sites wait for others to prove the billing model, adoption stalls in a feedback loop of caution.
In short, reimbursement friction is less a single binary risk and more a series of small, cumulative headwinds. But in a procedure-based platform, small headwinds add up quickly. Delcath must continue to push on all fronts, payer education, hospital engagement, coding optimization, and real-world evidence generation, to ensure reimbursement keeps pace with clinical demand.
Indication expansion risk: Clinical, operational, and regulatory complexity in scaling beyond mUM
While metastatic uveal melanoma (mUM) provides a compelling strategic entry point for Delcath, the long-term success of the platform depends heavily on its ability to expand into larger, more commercially significant indications. These include colorectal cancer liver metastases (CRLM), hepatocellular carcinoma (HCC), and intrahepatic cholangiocarcinoma (iCCA), among others.
Each of these indications offers materially higher patient volumes and market potential, but brings with it added complexity, competition, and clinical risk. The transition from mUM to these broader markets is not guaranteed, and represents a critical inflection point in the investment thesis.
The first layer of risk is clinical translation. While HEPZATO's mechanism - regional, high-dose, liver-isolated chemotherapy - is conceptually well suited for multiple hepatic-dominant cancers, the response dynamics, toxicity profiles, and therapeutic alternatives vary considerably across tumor types. For instance, CRLM patients are often heavily pretreated, and their disease can exhibit more heterogeneous vascularity and drug resistance than mUM.
Similarly, HCC patients frequently present with compromised liver function due to underlying cirrhosis, making them more vulnerable to hepatic toxicity even in an isolated perfusion setting. This raises the stakes for dosing, filtration efficiency, and patient selection. What works in mUM, a relatively homogeneous, liver-centric, otherwise systemically manageable disease, may not translate cleanly to more biologically or anatomically complex contexts.
Delcath’s CRLM program, for example, is progressing under an FDA-mandated randomized trial design, rather than the single-arm structure that was acceptable in mUM. This raises the bar for statistical significance, trial enrollment, and regulatory clarity. CRLM also intersects with a much more competitive therapeutic landscape, including Lonsurf, Stivarga, FOLFIRI, and evolving immunotherapy combinations, which means Delcath will need to show not just efficacy, but differentiated value against entrenched options. The risk is not simply clinical failure, but clinical ambiguity: results that show activity but fall short of definitively shifting standard of care.
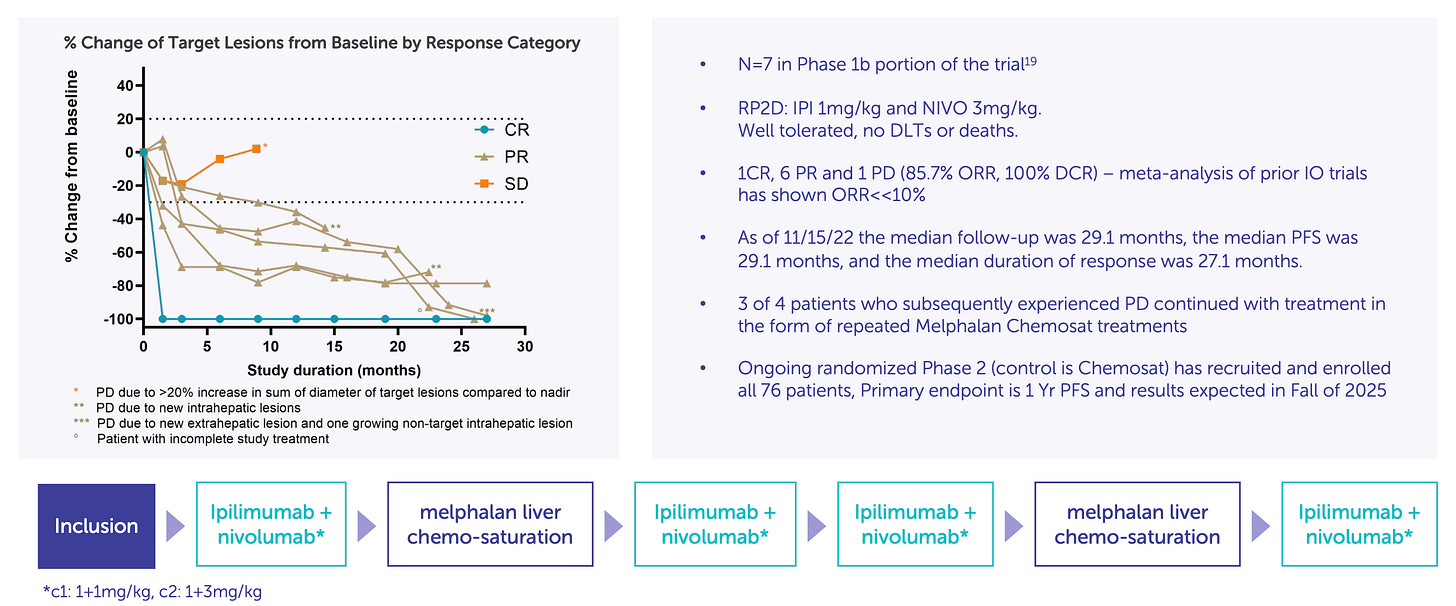
Second, there is a regulatory risk. While Delcath benefited from orphan drug designation and a sympathetic review environment in mUM, broader indications like CRLM or HCC will not have that same latitude. These are diseases with multiple approved therapies, active clinical pipelines, and entrenched stakeholders. The FDA’s tolerance for risk is lower, and trial expectations are higher.
Any delays in protocol acceptance, patient enrollment, or data readouts could materially push back the timeline for supplemental NDA filings. Moreover, if a confirmatory trial is required, as is likely for CRLM, the capital burden increases, and timeline to monetization extends significantly. This slows down the flywheel effect investors are counting on: site activation for mUM leading to volume expansion across multiple indications.
Third, the commercial dynamics change. Unlike mUM, where patients cluster in a small number of referral centers, CRLM and HCC are managed across a more distributed care network. This requires broader site activation, potentially beyond academic hubs, and introduces new logistical and training challenges.
It also increases Delcath’s exposure to variability in physician behavior, some of whom may not be familiar with perfusion-based delivery or may prefer more familiar systemic agents, even if they’re less effective in liver-dominant settings. The procedural commitment required of centers remains the same, but the commercial terrain becomes less centralized, more competitive, and more dependent on regional advocacy and payer cooperation.
Finally, there is an investor psychology risk embedded in this transition. mUM’s rarity and unmet need made it a logical, and forgiving, entry point. The investment community may not penalize early growing pains there. But once Delcath begins to speak seriously about expanding into large, high-profile oncology markets, expectations will rise quickly.
A moderate delay, an underwhelming Phase 2 result, or a cautious FDA response could trigger disproportionate negative sentiment, even if the long-term thesis remains intact. The market may shift from seeing Delcath as a differentiated rare-disease solution to judging it by blockbuster oncology metrics, a dangerous mismatch unless expectations are carefully managed.
Indication expansion is the core of the company’s forward value creation, but it is also the point at which execution risk sharply increases. The company will need to simultaneously manage more complex trial designs, broader patient heterogeneity, payer engagement in more nuanced cost-benefit environments, and physician education beyond its initial set of champions. Expansion isn’t optional, it's a strategic necessity. But its path is not linear, and any missteps will ripple through adoption curves, valuation, and investor confidence.
Procedural adoption curve: Overcoming behavioral friction in the standardization of novel oncology techniques
In many respects, the greatest long-term threat to Delcath is not scientific obsolescence, pricing pressure, or regulatory rejection, it is clinical apathy. Even with an approved product and strong early data, a procedural oncology platform like HEPZATO cannot succeed without sustained, repeatable behavior change across multidisciplinary hospital teams.
Unlike a drug, which a physician can prescribe with minimal operational disruption, HEPZATO requires institutional choreography: scheduling IR suite time, aligning anesthesiology, preparing perfusion equipment, and managing inpatient recovery. The resulting challenge is not just logistical, it is cultural. And that makes the adoption curve inherently non-linear.
In healthcare systems, especially large academic centers, new procedures compete for attention in crowded, highly constrained environments. Scheduling windows are limited, clinical staff are overextended, and administrative burden is rising. Unless a new procedure offers overwhelming clinical or economic incentive, it risks being deprioritized, especially when internal champions rotate out, leadership changes, or short-term complications arise. This said, clinical data success is well above-average in Delcath.
HEPZATO’s complexity, while defensible from a moat perspective, simultaneously raises its threshold for routine adoption. It must not only demonstrate value, it must become easier to perform with every iteration, or risk falling into the category of "interesting but impractical."
This is not a theoretical concern. The medical device industry is replete with precedents of novel procedures that delivered strong outcomes but failed to scale due to adoption bottlenecks. Consider intraoperative radiation therapy, laparoscopic robotic approaches before Intuitive Surgical, or even regional chemotherapy methods like isolated limb perfusion: all technically sound, all initially promising, and all hampered by inconsistent adoption because they demanded too much change from already stressed systems. Delcath must avoid this fate. It must not only win FDA approval, it must create institutional muscle memory.
This risk is magnified by the limited procedural TAM at each site in early indications like mUM. Even at top centers, the number of eligible patients may be low, often in the single digits per quarter. This creates a dangerous dynamic: if sites go weeks or months without performing a HEPZATO procedure, clinical teams lose familiarity, anesthesiology becomes hesitant, inventory coordination weakens, and enthusiasm wanes.
The therapy becomes “known, but dormant.” Unless Delcath can layer new indications onto existing infrastructure, or drive deeper throughput per site, early adoption could plateau well before it becomes self-sustaining.
To its credit, Delcath is actively working to compress this curve. The company has built out a procedural support infrastructure, including field training teams, onboarding specialists, reimbursement liaisons, and operational playbooks tailored to institutional needs.
It is also focusing early launches on high-volume, centralized institutions with known capacity for interventional trials and procedural innovation. But even with these supports, the risk remains: if the procedure does not become routinized within the first 6-12 months at a given center, it may be relegated to last-resort status or dropped entirely.
Another risk is that key stakeholders within the institution may not align. A highly motivated interventional radiologist may push for adoption, but if medical oncology, anesthesia, or nursing staff resist, due to perceived complexity, resource constraints, or lack of training, institutional buy-in falters.
These silos are hard to break down without strong internal champions and clear economic rationale. Delcath must effectively sell not just the clinical efficacy, but the institutional value proposition to multiple verticals within each center, a much more complex sale than traditional oncology drugs face.
Finally, there’s the broader systemic issue of evidence hierarchy and inertia in guidelines. Even if HEPZATO is clinically effective, it may take years before it is embedded in NCCN guidelines, widely reimbursed off-label, and integrated into standard-of-care pathways. Until then, physicians may be reluctant to refer patients, administrators may hesitate to allocate resources, and payers may scrutinize claims. This creates a temporal lag between early success and broader adoption, a lag that can undermine investor confidence unless carefully managed and transparently communicated.
The adoption curve for Delcath is not just about scientific data, it’s about behavioral economics within hospitals. Every step of the procedure must become more intuitive, every friction point must be sanded down, and every stakeholder must see value. If the company succeeds in this, it could establish a procedural franchise akin to TAVR or robotic surgery. If not, it risks becoming another technically sound but operationally fragile oncology tool.
Capital constraints and dilution risk: Balancing platform ambition with fiscal discipline
Delcath’s business model, a procedural oncology platform targeting multiple solid tumor indications, is operationally and clinically intensive. It demands not only robust clinical data, but also significant investments in hospital activation, field support, supply chain logistics, and regulatory engagement across jurisdictions.
While this capital intensity is justified by the long-term leverage of its delivery infrastructure, it introduces an inescapable strategic tradeoff: the company must balance multi-year strategic execution with the financial reality of limited cash reserves and a still-immature revenue base. In this context, capital constraints and dilution risk emerge as a core consideration for long-term investors.
As of its most recent disclosures, Delcath is not yet cash-flow positive. It remains reliant on external capital to fund its expansion into new indications, scale commercial operations, and potentially conduct confirmatory trials required for broader label expansion. While early mUM revenue may begin to offset burn modestly, it is unlikely to fully fund the company’s ambitions for several years.
This places the company in a familiar, but precarious, position seen across emerging medtech and biotech platforms: dependent on the capital markets for runway, but punished by them for needing more.
What distinguishes Delcath from many high-burn, pre-revenue biotech companies is its apparent capital discipline. Management has historically raised capital around meaningful milestones - such as trial progress, regulatory approval, or commercial inflection - rather than on a blind runway-extension basis. Additionally, it has kept its operating footprint lean, built a commercial team with focused scope, and resisted the temptation to prematurely expand into tangential R&D areas. This suggests a leadership team that understands the balance between ambition and solvency, a positive signal in an industry often plagued by dilutionary excess.
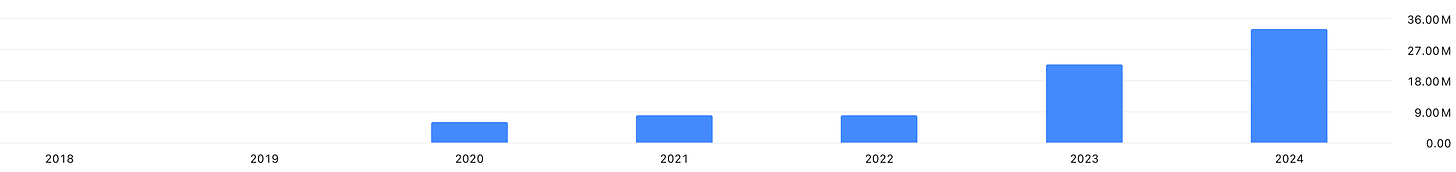
Nonetheless, dilution risk is real and material. If early commercial traction is slower than expected, whether due to onboarding delays, reimbursement lags, or clinical inertia, the company may be forced to raise capital at depressed valuations, significantly impacting long-term shareholder returns.
Given the relatively modest institutional ownership and limited float, even small equity raises can have outsized effects on share price volatility. This risk is compounded by the potential need to finance new indication trials, international expansion, and inventory buildup for scaled procedures, all of which require capital before they produce meaningful revenue.
One potential mitigant to dilution is non-dilutive financing. As Delcath moves into commercially validated territory, it may unlock opportunities for debt financing, royalty-based deals, or ex-U.S. licensing partnerships that reduce reliance on equity markets. Additionally, the company’s strategic focus on a capital-efficient expansion, through modular site activation and repeat procedure throughput rather than large sales force expansion, helps reduce variable cash burn over time. If executed well, these levers could extend the runway without compromising ownership structure.
Importantly, the nature of dilution risk is also tied to investor expectations. For shareholders with a multi-year view, dilution that funds clear commercial expansion or accelerates indication breadth may be accretive in long-term value terms, even if it is temporarily dilutive in share count. The problem arises when dilution is driven by weak execution, avoidable delays, or capital misallocation, scenarios that undermine the core thesis rather than extend it. To this point, Delcath has not shown signs of fiscal irresponsibility, but its capital stack and liquidity position must be monitored closely.
The firm operates at the intersection of procedural complexity and platform ambition, a model that is inherently capital-hungry in the near term, but potentially self-funding in the long term. The challenge for investors is to underwrite the company’s financial trajectory alongside its clinical one.
If commercial execution matches plan and indication expansion proceeds on schedule, future raises may be strategic, not survival-driven. But if traction lags or markets tighten, the company could face valuation pressure precisely when access to capital is most needed. The risk, in short, is not dilution itself, but dilution without leverage.
Delcath’s path to market dominance is paved with complex, yet navigable, hurdles. It has the clinical data, strategic intent, and organizational alignment to succeed, but those assets must now translate into throughput, reimbursement clarity, and scalable hospital behavior.
Unlike molecular biotechs, this is not a binary bet on trial data; it’s a multi-variable bet on procedural adoption, system integration, and capital-efficient execution. The reward is a durable platform with wide indication leverage, but the risk is delay, dilution, or drift. For investors, the thesis isn’t whether HEPZATO works. It’s whether Delcath can make it work everywhere, fast enough, with the capital it has.
Many of these risks are amplified, or tempered, by the broader structural dynamics of the biotech business model.
12. Biotech Operating Context
Delcath doesn’t just need to prove its science, it needs to out-execute a fragmented healthcare system, capitalize on infrastructure compounding, and win a long game in a market wired for short-term attention.
Delcath operates at the crossroads of two asymmetries: the binary, high-stakes economics of biotech, and the procedural inertia of interventional medicine. While its platform sits within oncology, its path to value creation doesn’t resemble traditional biotech’s molecule-driven model. Instead, it hinges on infrastructure leverage, behavior change, and compounding procedural capacity, an execution marathon in a market addicted to sprints.
Structural asymmetry: How biotech businesses compound or collapse
Biotech as a sector is defined by its extremes. Companies are often binary: they either create enormous value by achieving regulatory approval, reimbursement, and adoption, or they burn through capital and dissolve with little to no salvageable value.
The reason for this structural asymmetry lies in the non-linear payoff profile of biotechnology: R&D costs are front-loaded, regulatory timelines are long, and revenue is often back-ended and contingent on multiple system-level approvals. Yet when these barriers are overcome, the rewards can be massive: high-margin revenue, long exclusivity periods, and category leadership.
Delcath fits this profile in certain ways, but differs in others. Unlike traditional biotech firms built around molecular discovery, Delcath’s product is procedural, not pharmaceutical. The HEPZATO KIT is a physical intervention, a drug-device combination that delivers chemotherapeutic agents through hepatic perfusion, managed by interventional radiologists.
This distinction matters: while many biotech companies rely on scientific novelty to drive value, Delcath relies on institutional adoption and procedural standardization. Its risk profile is therefore less about scientific failure, and more about execution, adoption, and system integration.
At the same time, Delcath retains many of biotech’s financial characteristics. It required years of capital-intensive development with no revenue, was unprofitable throughout its clinical stage, and still depends on the capital markets for runway. Its revenue curve, like many biotech firms, is expected to be J-shaped, with a long flat tail of setup followed by potential rapid scaling if adoption accelerates. This is the classic biotech compounding curve, but it must be earned through operational excellence, not simply clinical endpoints.
For investors, this creates a hybrid valuation logic. Delcath cannot be valued like a pure medical device company with immediate, linear revenue. Nor can it be modeled like a biotech firm with speculative pipeline multiples based on platform science. Its value is tethered to something harder to quantify: the speed at which it can turn clinical approval into institutional routine, and the breadth of hepatic oncology it can ultimately capture. The upside is real, but it is gated by process, not just science.

Ultimately, Delcath embodies the bifurcated nature of biotech: if it wins, it wins big, not just in mUM, but in CRLM, HCC, and other indications that follow. If it fails to cross the operational chasm, its assets will have little residual value. There is no middle ground. This structural truth is not unique to Delcath, it defines the business model of biotech at large. But Delcath’s blend of engineering, oncology, and procedure-based reimbursement makes that asymmetry even more pronounced. Investors must therefore underwrite not just the product, but the infrastructure required to make that product matter.
Time horizons and the market’s attention deficit
Biotech companies rarely fail because their core science is flawed. More often, they fail because they cannot sustain institutional commitment, investor interest, or operational discipline over the multi-year grind it takes to commercialize a therapy. The mismatch between business timelines and market expectations is a structural feature of the sector, and it disproportionately punishes companies like Delcath, whose models rely on execution, education, and infrastructure-building rather than quick inflection points.
In Delcath’s case, the market’s attention span works against the company’s business logic. HEPZATO KIT has been approved for mUM, a milestone that de-risks clinical efficacy and regulatory acceptance. But the next phase of the company’s value creation, expanding site adoption, standardizing procedural workflows, and growing indication breadth, is inherently slow, cumulative, and unflashy.
It is not the kind of story that lends itself to quarterly “beats.” Instead, it is a multi-year narrative of system integration, institutional behavior change, and revenue curve steepening over time. This is not a story most short-horizon funds are willing or able to underwrite.
The result is predictable: Delcath trades at a valuation disconnected from its potential cash flow leverage. Investors anchored to typical biotech milestones, e.g., fast-following pivotal readouts, licensing deals, or early buyout speculation, will find Delcath's trajectory underwhelming. Its news flow is operational, not scientific.
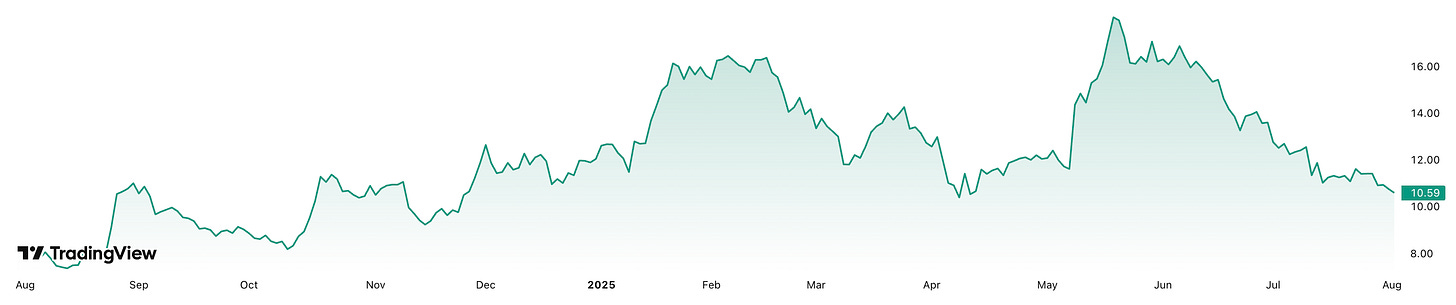
Its success depends on how many hospitals complete training, how many procedures are performed per center per quarter, and how efficiently it can expand its indication base. These are real business levers, but they are structurally ignored by the market until revenue shows up on the income statement.
Ironically, this misalignment creates opportunity for long-term investors. In a sector dominated by option-like trades on binary events (trial readouts, CRLs, M&A speculation), Delcath represents something different: a durable, executable operating story, with known infrastructure needs, clear reimbursement frameworks, and a methodical path to value creation.
But that path requires patience, and most market participants are structurally unwilling to wait. Institutional PMs are evaluated quarterly. Retail investors chase momentum. And even dedicated healthcare funds often reallocate capital every few months based on shifting pipeline sentiment.
This market attention deficit has real consequences. It can depress valuation, restrict access to low-cost capital, and impair the company’s ability to recruit long-term shareholders who can support multi-cycle execution. Delcath must navigate this pressure carefully, choosing when to communicate progress, how to stage capital raises, and how to protect strategic initiatives from short-term distractions. Management’s ability to keep internal alignment in the face of external impatience will be critical.
For investors with true long-duration capital, however, this mismatch is not a problem, it’s an edge. It allows entry into the story before platform leverage is visible in revenue or EBITDA. It offers exposure to a unique kind of compounding, not in molecules or market caps, but in institutional relationships, procedural repeatability, and multi-indication expansion from a shared infrastructure base. The market is impatient. Delcath’s business cannot be. That tension defines the opportunity.
Platform optionality vs. operational focus: Walking the line between vision and execution
One of the defining traits, and strategic tensions, in the biotech sector is the interplay between platform optionality and operational focus. The most attractive investment cases are often built on technologies that can be extended across multiple diseases, modalities, or use cases. Investors love platform narratives because they imply long-term leverage, R&D efficiency, and category leadership. Yet the irony is that platform companies often fail not due to scientific limits, but due to executional distraction, spreading themselves too thin before proving success in a single domain.
Delcath faces this exact tension. Its underlying technology, regionalized high-dose chemotherapy with systemic isolation via percutaneous hepatic perfusion (PHP), is not indication-specific. In principle, it can be deployed across a wide range of liver-dominant cancers, including CRLM, HCC, and iCCA.
Moreover, with sufficient procedural infrastructure in place, the marginal cost of indication expansion is relatively low: the same hospitals, delivery system, and clinical personnel can serve multiple tumor types. From a platform perspective, this offers remarkable capital efficiency.
But the opportunity to go wide must be balanced with the need to go deep, to operationally saturate a single use case before scaling to others. This is where many biotech companies falter. Instead of building momentum in one clinical or commercial domain, they chase too many indications, spread R&D dollars across heterogeneous trials, and dilute commercial focus. The result is “optionality without gravity”, a pipeline rich in press releases but poor in execution.
Delcath has thus far resisted this trap. Its current approach demonstrates disciplined sequencing: first, build clinical credibility and procedural infrastructure through mUM, a rare disease with a high unmet need and a streamlined path to approval. Second, leverage that installed base to expand into CRLM, a more complex but commercially meaningful market.
Third, explore ex-U.S. partnerships to drive regional scale without distracting internal bandwidth. Importantly, the company is not yet pursuing immunotherapy combinations, device licensing, or early-stage liver indications that could fragment its narrative. It is focused, and that focus is strategic.
Still, the temptation to overextend remains. As hospitals begin using the system, pressure may mount to “do more” with the platform, especially if early commercial traction underwhelms. Management must resist the allure of premature expansion, instead maintaining clarity of sequencing and evidence depth. A single procedural platform can support multiple billion-dollar indications, but only if it becomes clinically embedded first.
For investors, this balance is key. The platform thesis is real: the technology has physical, clinical, and economic modularity. But that optionality is not the core near-term driver, it is the upside tail. What matters now is commercial lock-in and throughput scaling in mUM, followed by CRLM. The platform becomes valuable only once the base case is proven. Until then, any claim of breadth must be interpreted not as growth, but as potential energy waiting for validation.
From science to infrastructure: The hidden leverage in procedural oncology
Most early-stage biotech companies are built on molecules, ideas made real through clinical validation and regulatory approval. But molecules, even great ones, rarely come with infrastructure. They rely on existing delivery mechanisms (e.g., oral, IV, subcutaneous) and existing physician behavior.
As a result, even groundbreaking drugs often fall into the same commercial mold: market access, sales force coverage, pricing strategy. Procedural oncology, by contrast, is not just about efficacy, it’s about embedding a new mode of therapy inside a complex delivery ecosystem. And while this raises the initial burden, it also unlocks a very different kind of leverage.
Delcath is not just launching a drug, it's deploying a platform that fundamentally alters the delivery of high-dose chemotherapy. Once a hospital is trained, credentialed, and operationally integrated with the HEPZATO KIT procedure, that infrastructure is durable and reusable. Every subsequent procedure, whether for mUM, CRLM, or other indications, benefits from that initial setup investment.
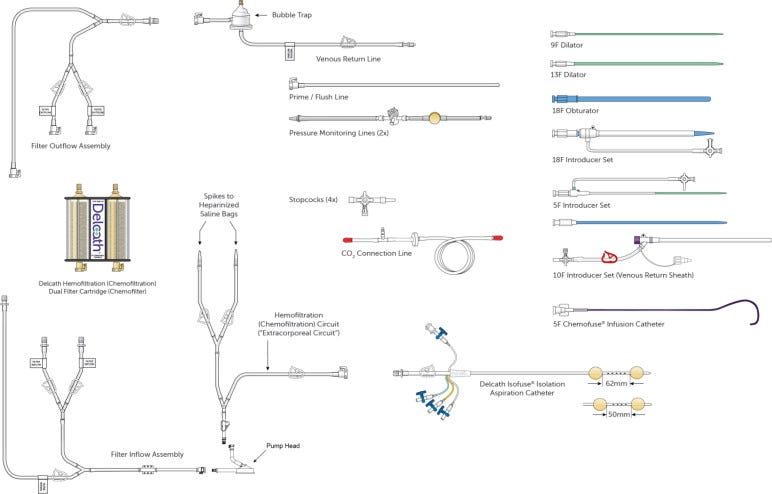
This is the inverse of traditional biotech, where each new drug requires a separate set of prescriber education, distribution agreements, payer negotiations, and post-marketing studies. Delcath builds once and scales across multiple revenue streams within the same installed base.
This is the essence of infrastructure leverage, the ability to generate compounding value not by discovering more molecules, but by reusing procedural capacity across a growing TAM. Once HEPZATO becomes standard practice in treating liver metastases at a top cancer center, that center can extend its use to other indications with relatively low marginal effort. Every new physician onboarded, every nurse trained, every billing pathway cleared becomes a reusable asset. The business model begins to resemble that of surgical platforms or interventional devices, not small-molecule pharma.
But this leverage must be earned. Infrastructure becomes an asset only once it is activated, standardized, and integrated into routine practice. Delcath must build the playbooks, training protocols, billing support systems, and physician advocacy tools to make the procedure repeatable and dependable.
The procedure has to move from “interesting” to “routine.” Once it does, the leverage flips: new indications become incremental volume, not fresh launches. The ROI per trial, per approval, per patient increases. And the company’s commercial cost per dollar of revenue falls.
This is a different kind of biotech compounding, one based not on molecular pipelines, but on clinical repetition and procedural standardization. It's what made companies like Intuitive Surgical dominant: not the robot itself, but the hospital’s commitment to using it as the default approach across procedures. Delcath has the opportunity to do something similar in hepatic oncology. But doing so requires patience, sequencing, and operational excellence.
For long-term investors, this shift from “science risk” to “infrastructure leverage” is a central part of the thesis. Delcath's early years were about proving the therapy. Its next phase is about building the delivery system. Once that system is in place, and trusted, every new indication is not a reinvention, but an extension of infrastructure that already works.
Delcath’s opportunity is structurally rare: a validated procedural platform with multi-indication potential, defensible reimbursement pathways, and repeatable institutional value. But the playbook isn’t biotech’s usual hit-or-miss sprint, it’s a multi-year compounding loop built on hospital adoption, procedural throughput, and smart sequencing.
The company’s future depends less on its pipeline and more on its patience, discipline, and ability to build something the market won’t fully value until it’s already working.
This context is only as precise as the financial engine supporting them, capital strategy now takes center stage.
13. Financial Profile & Capital Allocation
Delcath isn’t burning capital to survive anymore, it’s building operating leverage into a procedural platform with real pricing power and expanding gross margins.
After years of high burn and dilution typical of early-stage biotech, Delcath’s financial story is pivoting, from speculative science to real revenue, margin leverage, and disciplined capital deployment. The recent commercial traction of HEPZATO has transformed the company’s financial complexion, offering investors a visible line of sight to cash flow breakeven and a capital strategy rooted in platform scale, not survival mode.
Revenue evolution and gross margin inflection: From zero to signal in a niche launch
Delcath’s recent revenue trajectory marks a turning point in the company’s long development arc, offering long-term investors a first glimpse of platform monetization at commercial scale. After years of near-zero product sales, Delcath reported LTM revenues of $53.85 million, up from just $2.07 million in 2023, a staggering ~25x increase year over year.
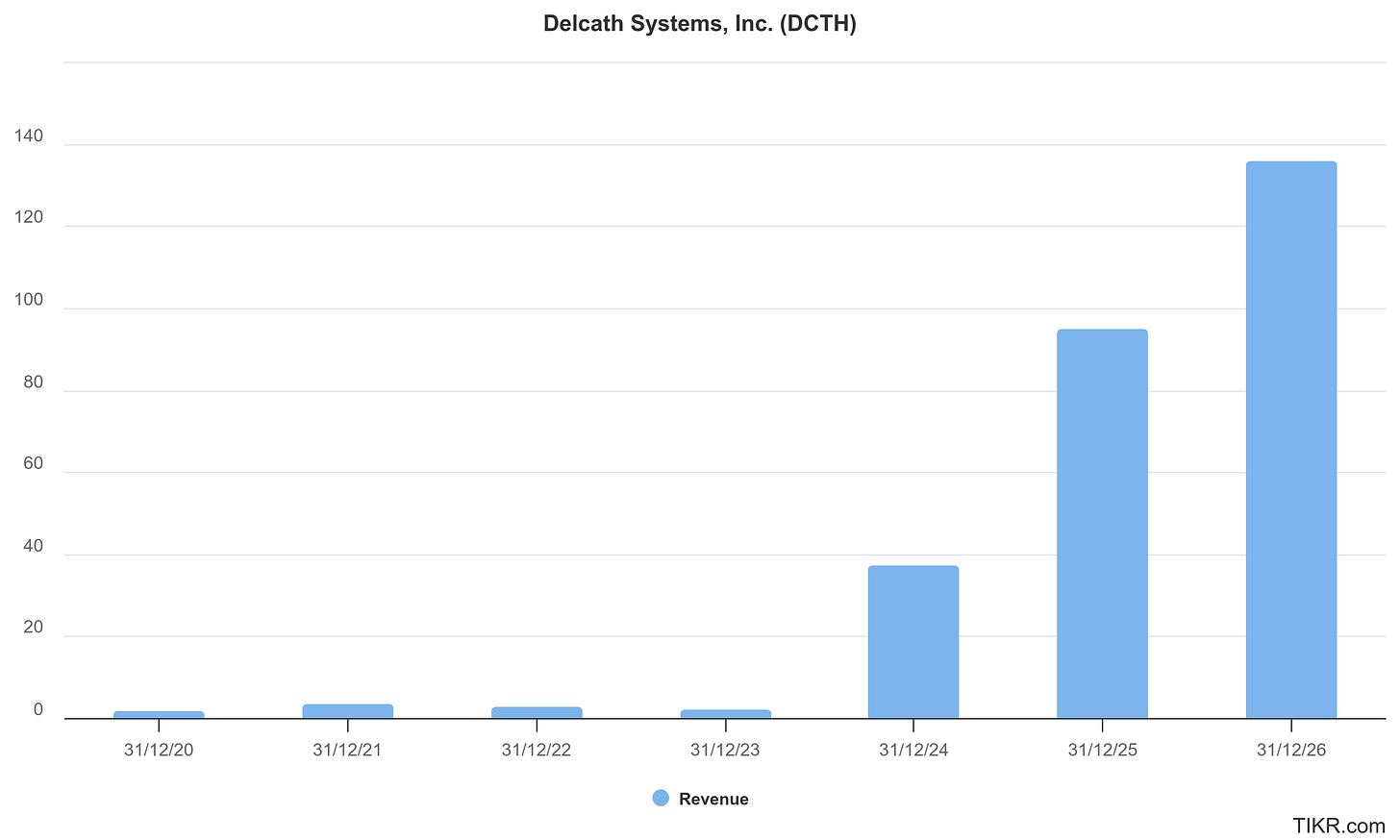
This growth is not the product of licensing fees or one-off milestone payments, but rather real-world procedural revenue tied to increasing utilization of the HEPZATO Kit for metastatic uveal melanoma (mUM) in U.S. cancer centers. This dynamic matters deeply: it shifts the company from a speculative, pre-revenue biotech to an early-stage commercial franchise with tangible evidence of hospital adoption, reimbursement integrity, and clinical demand.
Crucially, this revenue is tightly linked to procedural throughput, not pure drug volume, meaning every incremental patient reflects both demand for the therapy and the successful onboarding of complex hospital infrastructure. Unlike traditional biotech launches where vials can be shipped and shelved, Delcath’s revenue requires full procedural coordination: trained interventional radiologists, perfusionists, oncology support staff, and hospital billing systems.
That this infrastructure is now being used consistently in practice, and at a scale large enough to generate over $50 million in revenue, provides strong validation of the company’s operational execution and product-market fit in its first indication.
Moreover, the company’s gross margin trajectory reinforces the economic logic of its model. While high upfront CapEx and training costs would typically weigh on margins in the early launch phase, Delcath has already demonstrated substantial leverage: gross margins improved from 69.2% in 2023 to 84.9% in the latest reporting period. This inflection was driven by both scale efficiencies and product mix improvements.
The fixed costs associated with manufacturing, training, and distribution are now being absorbed across a growing volume of procedures. This signals the arrival of high-contribution-margin economics, where incremental revenue flows through with minimal friction. In biotech terms, this resembles a platform model more than a pure product play: revenue scales faster than cost, and margin expands with procedural density.
It is also important to note that this early revenue is concentrated in a small and well-defined patient population. With ~1,000-1,200 mUM patients in the U.S. eligible annually, Delcath’s initial commercial success reflects meaningful penetration of its first market.
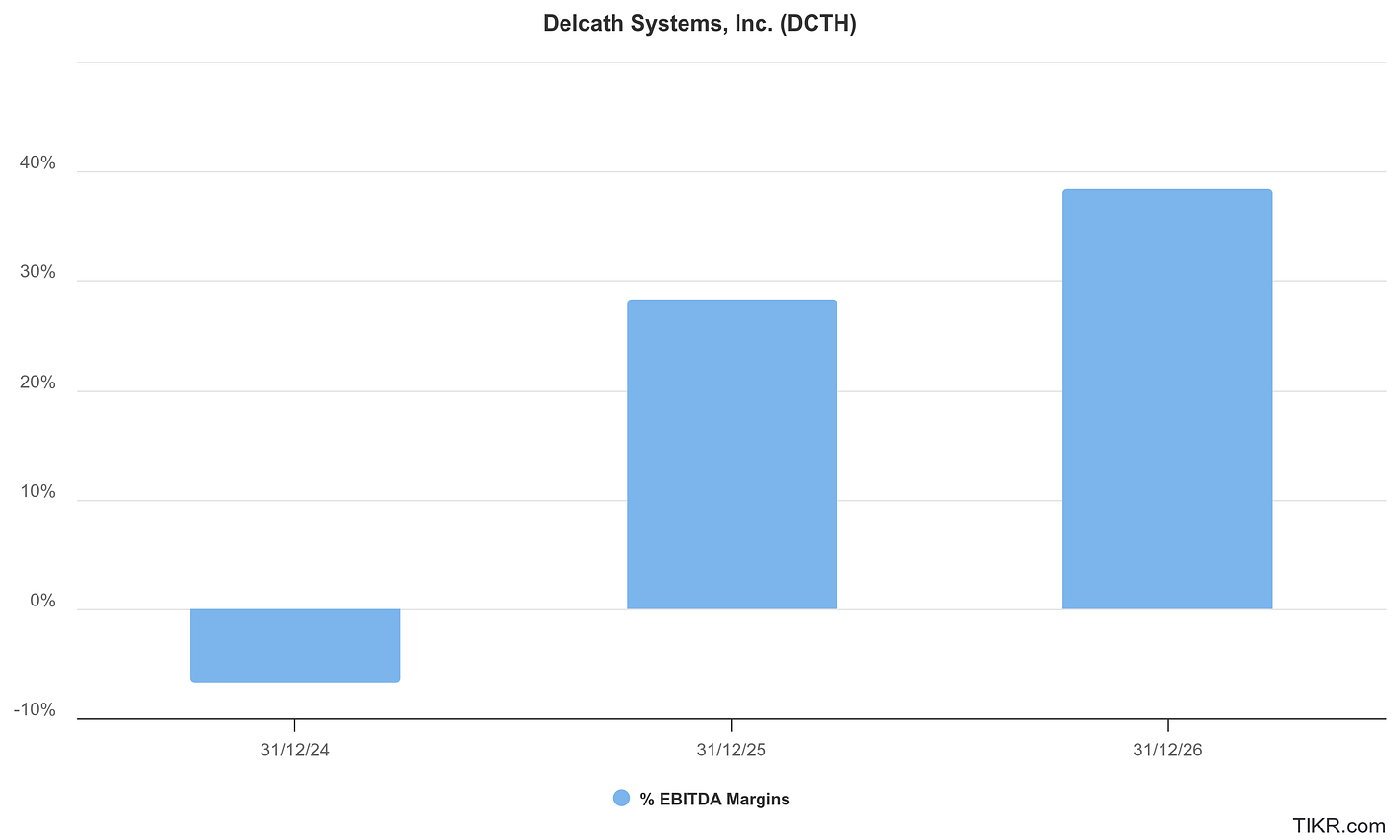
This offers investors three key insights: (1) the launch execution is real, not theoretical; (2) the early adopter sites are successfully delivering the procedure; and (3) the model is translatable to other concentrated hepatic tumors, where similar delivery logistics apply. Each dollar of current revenue is thus more informative than in a commodity drug launch: it signals the scalability of a procedural oncology franchise.
Finally, the procedural nature of HEPZATO sales, involving consumables, device components, and high-complexity support, offers potential for recurring per-patient revenue at a premium price point. The per-procedure pricing, while not disclosed in full detail, likely ranges from $40,000-$50,000, based on CMS reimbursement levels.
Repeat procedures, as often required in mUM, increase revenue per patient and enhance predictability of center-level economics. This also sets the stage for volume-based acceleration as larger indications like CRLM come online. If Delcath can activate more centers while increasing per-site throughput, a trend already underway, the revenue trajectory should steepen significantly, while gross margins continue to expand toward a best-in-class profile.
For long-term investors, this phase marks the inflection point where the company transitions from being a capital consumer to a platform that can compound operating leverage and generate real cash flow, contingent on disciplined expansion and continued clinical execution. The revenue curve, while still early, now has a visible slope. And the margin profile suggests that Delcath is not building a niche tool, but rather a scalable, profitable business model with defensible procedural economics.
Cash burn, runway, and capital formation: A biotech in transition from dilution to durability
Delcath’s historical financial profile has reflected that of a typical development-stage biotech: steep cash burn, serial equity raises, and a balance sheet structured around survival rather than efficiency. But 2024 marks a pivotal transition year.
With commercial revenues now scaling rapidly, R&D expenses normalizing, and gross margins expanding, the company is entering a phase where operating losses are compressing, and the underlying cash engine begins to matter more than capital raises. For investors, this marks the transition from valuation as optionality to valuation as compounding, and makes balance sheet quality a critical lens through which to assess sustainability.
Historically, Delcath’s operating cash burn has been substantial: free cash flow was -$31.3 million in 2023, following multi-year trends of -$22-25 million annually. But the most recent LTM figure shows burn declining sharply to -$7.6 million, driven by a combination of higher revenues and greater capital efficiency. This signals the beginning of a structural transformation.
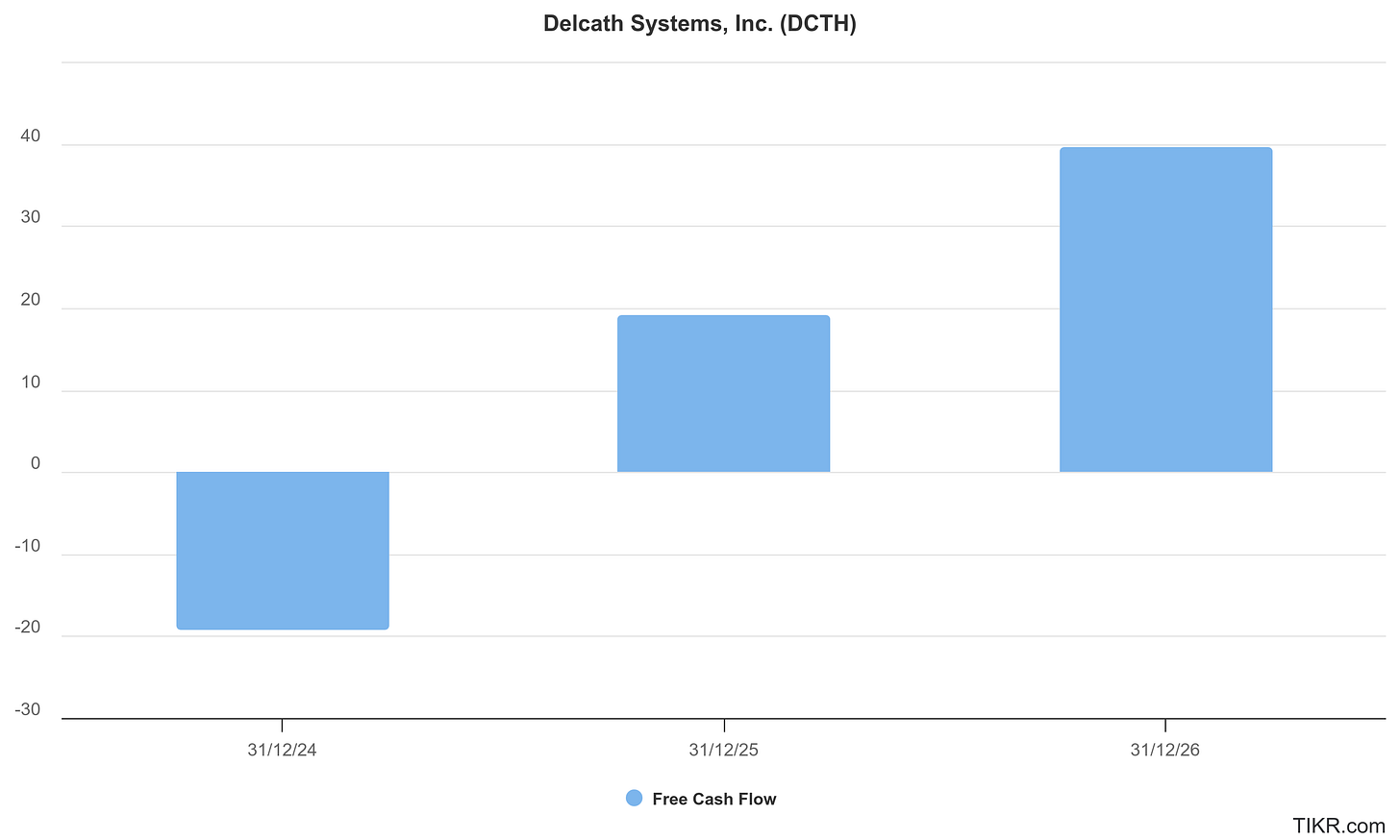
For the first time, product revenue is contributing meaningfully to fixed cost absorption, and the cash burden of running clinical trials and commercial operations is no longer open-ended. This dynamic suggests that Delcath may be within 12–18 months of breakeven cash flow if commercial momentum and margin expansion continue along their current trajectory.
On the balance sheet, the company holds $58.9 million in total cash and short-term investments as of the latest reporting period. With LTM cash burn at just under $8 million, this implies a runway of over 24 months under conservative assumptions, and potentially longer if commercial acceleration outpaces hiring and R&D spend.
Crucially, this position is achieved without significant reliance on debt: net debt is negative ($-57.9M), and long-term liabilities are modest. The company’s capital structure is clean, with no toxic convertibles, no contingent milestones owed, and minimal preferred equity ($5.55M). This gives Delcath significant strategic flexibility, whether to fund future trials, build commercial infrastructure, or pursue geographic expansion.
That said, the company’s equity base has expanded rapidly over the last few years, growing from 2.9 million shares outstanding in 2020 to nearly 35 million today. Much of this dilution came at lower valuations, reflecting the timing mismatch between R&D needs and commercial validation. While painful for early holders, this dilution now sets the stage for operating leverage to benefit current and future investors, especially if the company can hold its SG&A flat while revenues ramp. The key risk now is execution, not survival.
Importantly, management has shown disciplined capital formation behavior. Rather than chasing value-destructive licensing deals or bloated pipelines, Delcath has consistently raised equity to fund high-probability, data-driven initiatives, namely, the Phase 3 FOCUS trial, CRLM studies, and the commercial buildout. It has avoided overhiring, refrained from empire-building, and maintained tight control of COGS despite the complexity of the procedure. The result is a leaner-than-expected operation with a credible path to self-sustaining cash flow within the medium term.
For long-term investors, this transition, from capital consumer to margin-generating platform, is central to the thesis. With the balance sheet de-risked, the gross margin profile validated, and cash burn compressing, Delcath now has the luxury of choosing how and when to deploy capital, rather than merely raising to survive.
The next round, if any, can be optional, strategic, and accretive, a far cry from the liquidity-driven raises of the past. As cash flow neutrality approaches, valuation multiples will shift from discounting survival to discounting operating leverage and durable platform value.
R&D and SG&A discipline in transition: Controlling the bridge from science to scale
Delcath’s cost profile over the last several years reflects a company that has evolved through distinct operational phases, from a science-heavy, pre-revenue biotech into a commercial-stage, execution-oriented platform company. The firm’s ability to modulate its R&D and SG&A spend through each of these phases, without losing focus or overextending, speaks to a disciplined management culture that prioritizes durability over optics.
This cost structure evolution is not just a back-office detail. For long-term investors, it is one of the clearest signals that Delcath is building for compounded margin expansion rather than simply riding a transient launch window.
On the R&D side, expenses peaked at $18.6 million in 2022, primarily driven by the completion of the FOCUS Phase 3 trial for metastatic uveal melanoma and the groundwork for CRLM indication expansion. Since then, R&D spend has trended lower, $17.5 million in 2023 and $15.2 million in the latest trailing twelve months (LTM), a signal that core development milestones have been achieved, and the company is entering a more focused and less capital-intensive phase.
This does not reflect a retreat from innovation, but rather a deliberate transition toward indication leverage, where the same clinical infrastructure (e.g., hepatic artery delivery) is reused across different liver cancers, minimizing marginal development costs.
Delcath’s R&D philosophy also reflects strategic restraint. Unlike many biotech peers, it has avoided sprawling exploratory programs, platform science distractions, or unnecessary preclinical spending. Instead, it has concentrated investment around indications with operational adjacency and clear commercial endpoints, a playbook reminiscent of disciplined oncology developers like Exelixis or ImmunoGen.
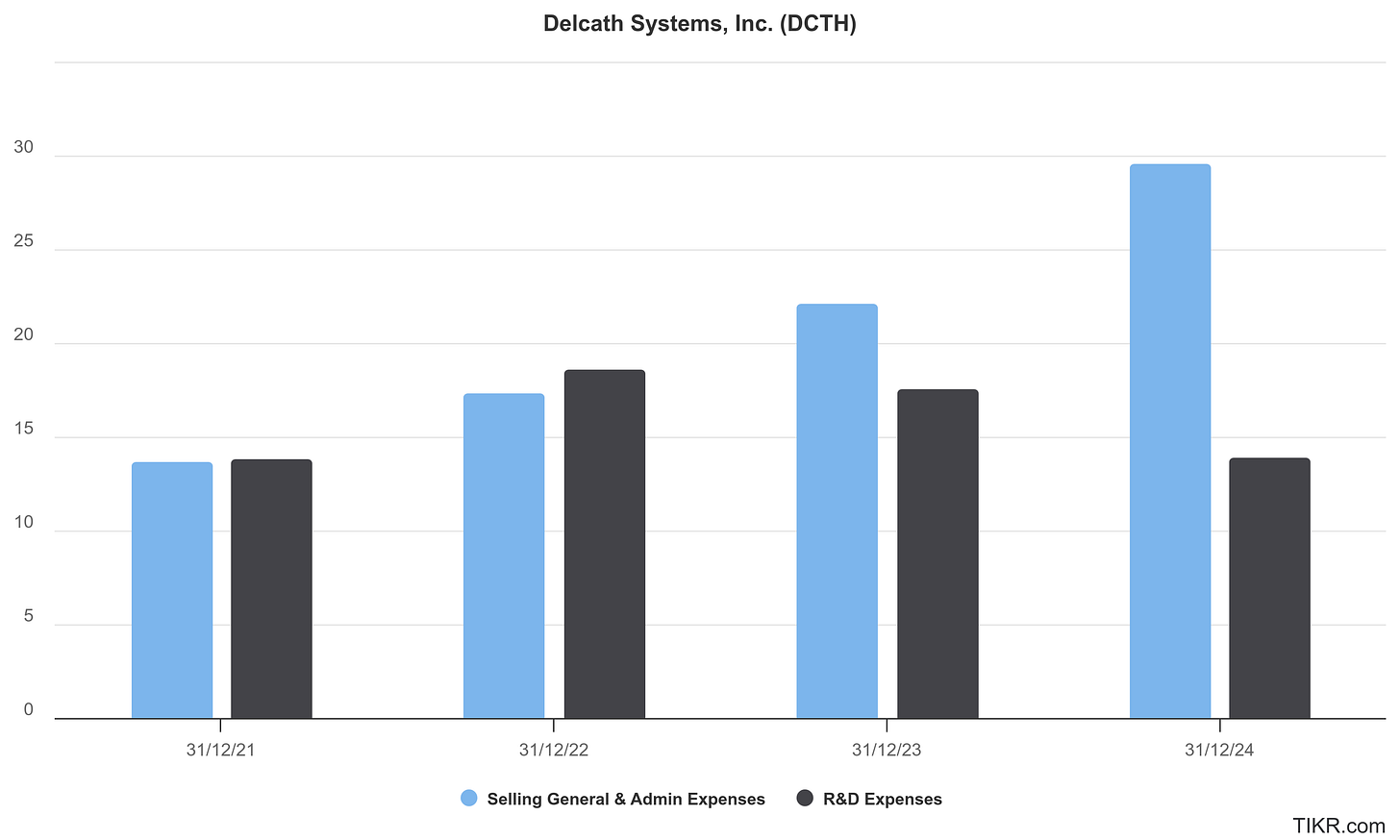
Each dollar of R&D is tied to a credible, FDA-trackable deliverable: either a pivotal trial (e.g., in CRLM), real-world safety studies (e.g., in HCC), or reimbursement infrastructure support (e.g., registry studies). This focus improves internal ROI and makes the path to profitability more predictable.
SG&A, meanwhile, has followed a logical upward slope consistent with commercial expansion. From $11.1 million in 2020, SG&A has grown steadily to $32 million LTM, tracking the buildout of the commercial field force, reimbursement team, and corporate infrastructure. But critically, this growth has not been exponential or bloated.
Even as revenue has scaled ~25x year-over-year, SG&A has risen at a measured pace, suggesting strong cost controls, lean headcount, and a culture of ROI-based spending. This is especially impressive for a company launching a high-complexity, hospital-based procedure, a setting where SG&A burn can balloon if unmanaged.
Looking ahead, Delcath’s SG&A is well-positioned to benefit from operating leverage. Many of the fixed costs, such as medical affairs, training infrastructure, and payer support, are already in place and can support multiple new indications without proportional increases in spend.
As more cancer centers are activated and procedural throughput per site increases, the marginal cost of commercial delivery will decline, driving EBIT improvement even in the absence of major top-line gains. This dynamic is rare in early-stage oncology and reflects the company’s procedural platform economics rather than a typical biotech sales model.
For long-term investors, the combination of declining R&D intensity and scalable SG&A is highly attractive. It not only signals proximity to cash flow breakeven, but also positions Delcath as a margin-expanding, platform-operating company rather than a one-shot drug developer. In a sector often plagued by fixed cost bloat and R&D vanity, Delcath’s fiscal discipline is a competitive advantage.
It suggests that capital raised in the future, if any, will not be used to plug structural inefficiencies, but to fuel strategic growth in a business already operating within tight cost control parameters.
Cost structure evolution and margin normalization: Leveraging fixed cost infrastructure for platform economics
Delcath’s evolving cost structure reflects a critical inflection point in its maturation from an R&D-heavy biotech into a leaner, margin-leveraging commercial platform. For investors, this evolution is not merely an accounting shift, it is a forward-looking indicator of whether the company can transition from perpetual dilution to durable operating cash flow.
With LTM financials now reflecting a sharp contraction in net loss, rising gross margins, and stabilizing OpEx growth, Delcath appears to be entering a phase of margin normalization, the phase where future revenue growth outpaces incremental cost.
The company’s gross margins are among the most important early signals. From 2020 to 2023, gross margins improved from 61.1% to 69.2%, but it’s in the most recent LTM period where leverage becomes striking: gross margin has expanded to 84.9%, indicating that a substantial portion of the fixed costs associated with manufacturing, delivery kits, and logistics have now been amortized across a growing base of procedures.
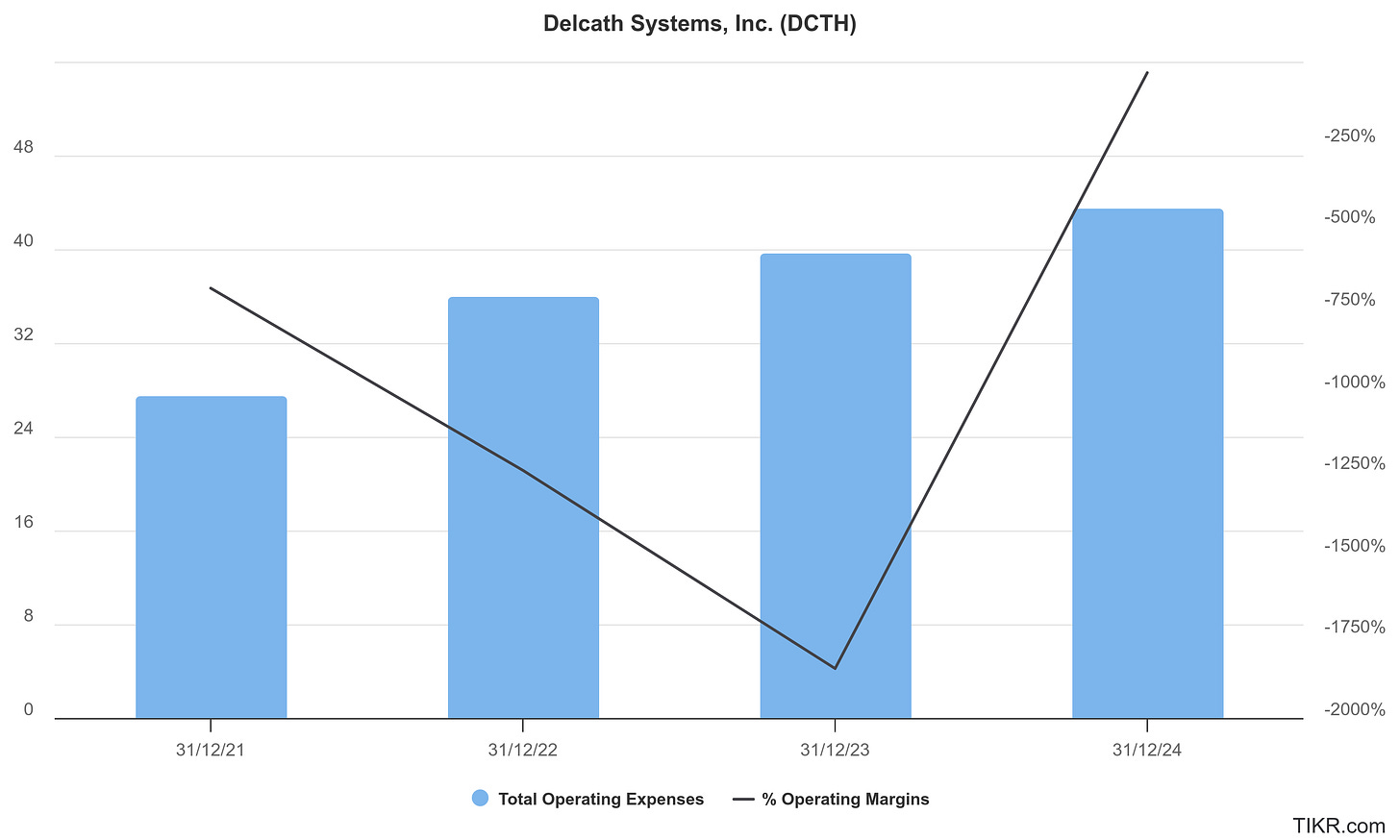
This is a significant achievement for a company whose product is not a simple vial of biologic, but a complex therapeutic kit involving drug, hardware, perfusion circuit, and specialist procedural training.
The key structural insight here is that Delcath’s platform exhibits fixed-cost leverage similar to that of a SaaS or surgical robotics company, where each new site or patient introduces relatively little incremental cost. Core components of the cost stack, production of HEPZATO (melphalan hydrochloride), kit assembly, device components, and distribution, all scale with volume.
More importantly, the procedural logistics, training burden, and payer onboarding are frontloaded, meaning new center activation becomes cheaper over time. These dynamics are rarely found in traditional biotech, and they position Delcath as a procedural platform with multi-indication upside, rather than a single-product oncology company.
On the OpEx side, we observe a stabilization trend that suggests spending discipline at scale. Total operating expenses (R&D + SG&A) rose from $22.3M in 2020 to $47.2M in the LTM period, but the majority of that increase occurred prior to revenue acceleration. SG&A has scaled to support commercialization, but R&D is already tapering as key programs reach maturity.
Importantly, the company is not layering in speculative research or unrelated indications, which helps contain spending and extend cash runway. As more indications are added and revenue grows, we expect OpEx as a percentage of revenue to fall materially, enabling EBITDA breakeven in the medium term without additional structural cost.
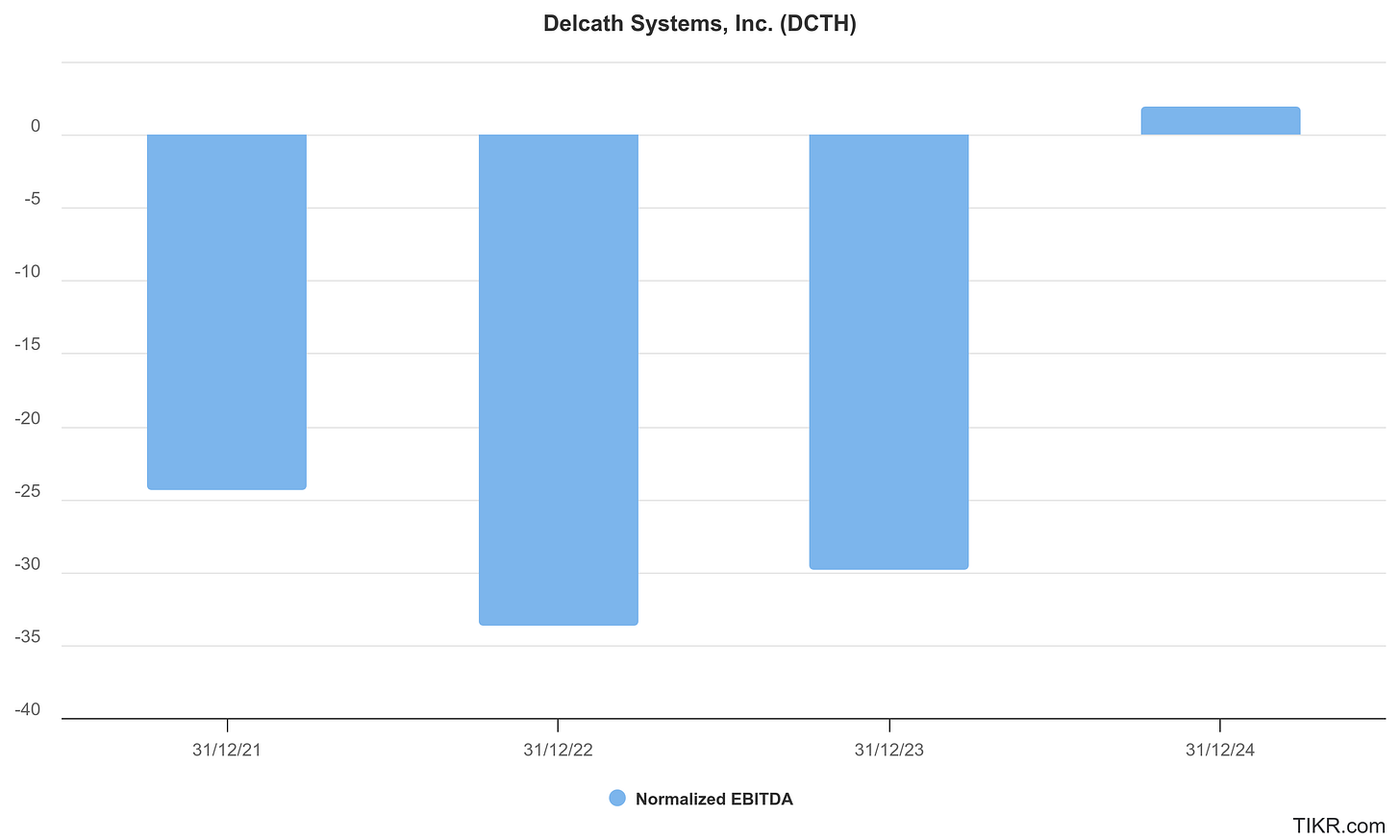
Looking forward, this margin normalization will likely accelerate. If the company can maintain high gross margins while pushing procedural volumes higher through center activation and CRLM expansion, fixed cost absorption will deepen.
Even without large revenue inflections, the existing commercial infrastructure can support operating leverage, creating margin expansion over time. Unlike traditional drug developers that rely on binary pipeline catalysts to swing toward profitability, Delcath’s path is procedural and cumulative: more patients, more throughput, same platform.
For long-term investors, the signal is clear. The company is no longer stuck in a high-burn holding pattern. It is building a business with scalable margins, operational control, and embedded leverage, where cost containment is not reactive, but strategic. This structure enables the business to grow without disproportionate capital needs, and it lays the foundation for Delcath to become a cash-generating oncology platform over time.
Capital allocation framework and strategic optionality: Deploying cash for scale, not survival
Delcath Systems’ approach to capital allocation is unusually disciplined for a company at its stage of maturity. While many emerging oncology players pursue growth at any cost, often leading to overexpansion, pipeline distraction, and bloated burn, Delcath’s recent trajectory reflects a leadership team that views capital not as an entitlement but as a scarce strategic resource.
The shift from survival-driven fundraising to selective, ROI-conscious capital deployment is now central to the long-term investment case. With a strengthened balance sheet, early commercial traction, and a tight operational focus, Delcath is positioned not just to conserve cash, but to strategically compound it.
As of the latest reporting period, Delcath held approximately $58.9 million in cash and short-term investments, with net debt deeply negative and no near-term financing pressure. This gives the company 24+ months of conservative runway at current burn levels, even before accounting for further revenue acceleration.
In effect, the company has earned the right to choose how and when to allocate capital, whether to fund expansion, develop the pipeline, or strengthen its procedural footprint. Unlike biotech peers who must finance every quarter’s R&D outlays with equity dilution, Delcath can now optimize for shareholder return and platform scale, not mere survival. The company’s capital allocation principles appear to rest on three key pillars:
Focused Indication Expansion: Capital is being directed toward clinically and operationally adjacent indications, such as colorectal cancer liver metastases (CRLM), hepatocellular carcinoma (HCC), and potentially intrahepatic cholangiocarcinoma. These are not speculative bets; they reuse the same delivery infrastructure, clinical playbook, and reimbursement model as the mUM launch. This approach ensures that every dollar of R&D spend leverages existing assets, minimizes marginal cost, and compounds platform value.
Commercial Infrastructure Scaling: Delcath is investing in site activation, training programs, and payer engagement, not bloated salesforces. This infrastructure is durable, reusable, and increasingly self-sustaining. The choice to keep SG&A growth in check, even while revenue has exploded, indicates a focus on long-term procedural throughput, rather than vanity metrics like headcount or surface-area coverage.
Avoidance of M&A Bloat: So far, Delcath has avoided the classic error of acquisition-driven distraction. The company has not pursued platform “bolt-ons,” pipeline-heavy assets, or corporate combinations that risk derailing focus or increasing overhead. While future tuck-in acquisitions may be on the table, particularly in interventional oncology tools or hepatic diagnostic technologies, there is no evidence of imminent or undisciplined M&A behavior.
Strategic optionality is also increasing. As revenue scales and cash burn compresses, Delcath could pursue selective international expansion, particularly in Western Europe where hepatic delivery infrastructure is mature.
Alternatively, the company could license rights to partners in Asia-Pacific markets, generating milestone revenue without straining internal bandwidth. On the capital structure side, management could consider non-dilutive financing (e.g. royalty-backed debt or milestone-based credit lines) to fund later-stage trials or commercial growth, especially if equity markets remain volatile.
Importantly, there is no pressure to act. With strong margins, expanding topline, and a modest cost base, Delcath can afford to be patient and wait for high-quality opportunities. The risk of forced, desperate capital allocation, a hallmark of many mid-cap biotechs, is low. And with its platform beginning to show compounding economics, future capital deployment has the potential to amplify shareholder value, rather than merely preserve runway.
For long-term investors, this sets up a very different capital trajectory than most emerging oncology companies. Delcath is not building for a quick sale or chasing a near-term pop. It is slowly assembling a high-margin, durable, procedural franchise that could operate independently, or eventually attract premium acquisition interest. In either case, capital allocation discipline is not a footnote. It is part of the moat.
Delcath is now operating from a position of control: revenue is rising, margins are expanding, and the balance sheet is healthy. With a lean cost structure, operational focus, and an infrastructure-light path to multi-indication expansion, the company is building a financial profile that can compound. In a sector too often defined by capital dependence, Delcath is quietly becoming a self-funding platform, and that changes everything.
With the financials mapped, the final piece is a long-range view of how this platform could evolve, and what success might look like.
14. Long-Term Strategic Outlook
Delcath isn’t just building a product, it’s laying the infrastructure for a procedural category that could redefine liver cancer treatment globally.
Delcath Systems is positioned at the confluence of three structural tailwinds: (1) the clinical need for liver-directed therapies in oncology, (2) the emergence of interventional oncology as a procedural discipline, and (3) the shift toward reimbursed, device-enabled treatment platforms that blend the scalability of medtech with the pricing power of oncology.
This section outlines how Delcath's strategy, platform architecture, and operating discipline set the stage for long-term structural expansion, whether or not the company remains independent. Rather than relying on binary inflection points, Delcath is executing a multi-year transition from product-centric biotech to platform-enabled procedural medicine, a transition that may ultimately redefine how advanced liver cancers are treated.
Procedural oncology as a structural shift: How Delcath is pioneering a new therapeutic modality
At the core of Delcath’s long-term thesis is the recognition that the company is not simply commercializing a drug or device, it is catalyzing a structural transformation in how liver-involved cancers are treated, with the potential to institutionalize a new class of interventional oncology procedures.
This shift is not incremental; it is categorical. Much like what Intuitive Surgical achieved with robot-assisted surgery or what TAVR platforms have done for valve replacement, Delcath’s HEPZATO platform aims to redefine the standard therapeutic delivery paradigm for select solid tumors confined to or predominantly located within the liver.
Today, liver-directed therapy remains an under-optimized corner of oncology. Systemic therapies, including chemotherapies and immunotherapies, often fail to deliver sufficient therapeutic concentrations to hepatic metastases due to poor perfusion and liver toxicity concerns. Conversely, local ablation or embolization procedures, while helpful for small lesions, are ill-suited for diffuse metastatic disease.
The market gap is clear: patients with high hepatic tumor burden but otherwise limited extrahepatic progression have no effective regional treatment that balances efficacy with systemic safety. Delcath’s Percutaneous Hepatic Perfusion (PHP) platform addresses this unmet need by enabling targeted, repeatable, high-dose drug delivery to the liver, while filtering systemic melphalan to mitigate toxicity.
What makes this thesis structurally different is the procedural model itself. PHP is not a product to be prescribed, it is a procedure to be performed, in specialized centers, by trained interventionalists. This model embeds Delcath’s platform within the workflows of tertiary cancer institutions, turning it into infrastructure rather than just a vendor product.
The company is thus positioning itself as a procedural gatekeeper for targeted hepatic drug delivery, a role that comes with strategic pricing power, high switching costs, and physician-level loyalty. Over time, if this procedural modality becomes standard of care across even a few liver-dominant cancers, Delcath could emerge as the platform-of-record for a category it is actively defining.
Critically, this thesis is not aspirational, it is already underway. In the U.S., top-tier academic centers have adopted the technology for mUM. CMS reimbursement codes have been established. Key opinion leaders are publishing real-world data. The product has entered European markets, albeit in early stages.
The long-term opportunity is to turn this early procedural footprint into a repeatable infrastructure layer, one that becomes the default for select cancers and potentially extends beyond oncology into areas like hepatic fibrosis, metabolic liver disease, or regenerative liver perfusion. The ambition is not just to treat more patients, it is to own the procedural rails for regional liver therapy. That is the true platform.
Indication sequencing as a compounding asset: Expanding the platform without reinventing it
A core strength of Delcath’s long-term strategy lies in the inherent modularity of its platform, enabling indication expansion without reinvention. Unlike traditional biotech companies that must revalidate scientific hypotheses, develop novel compounds, or shift biological targets to grow addressable markets, Delcath operates more like a therapeutic delivery infrastructure company.
Its value proposition is not confined to a single drug or cancer type, but to the delivery mechanism - a repeatable, validated system for administering high-dose chemotherapy to the liver, paired with a filtration circuit that reduces systemic toxicity.
This architecture allows Delcath to expand methodically across adjacent hepatic indications where the pathophysiology, delivery constraints, and treatment gaps align with the PHP platform. The company’s next expansion targets, colorectal cancer liver metastases (CRLM), hepatocellular carcinoma (HCC), and intrahepatic cholangiocarcinoma (iCCA), are not speculative.
Each of these tumor types has (1) high incidence of liver-dominant progression, (2) poor outcomes with systemic therapy alone, and (3) procedural compatibility with catheter-directed hepatic perfusion. That convergence reduces both clinical risk and commercial uncertainty.
Indication sequencing in this context becomes a platform-level flywheel. Each new indication shares the same core inputs: the melphalan drug payload, the HEPZATO kit, the PHP procedural protocol, and the training and support infrastructure already deployed at cancer centers.
From a cost and time-to-market standpoint, this means each expansion leverages existing capital, compresses marginal costs, and shortens regulatory timelines. From a commercial standpoint, it deepens center utilization, increases per-site revenue, and embeds Delcath more firmly in institutional workflows. This is not a new launch every time, it is a progressive expansion of throughput across the same rails.
The economic implications of this model are substantial. With modest incremental R&D, Delcath can unlock large new TAMs (e.g. CRLM alone affects 50,000+ patients per year in the U.S.), while achieving high incremental gross margins due to fixed platform costs. Unlike companies pursuing multi-asset pipelines that each require their own manufacturing, trial design, regulatory approvals, and commercial ramp, Delcath is layering value horizontally across a shared chassis.
This produces a compounding dynamic: more indications drive more procedures per center, which increases product pull-through, accelerates payor adoption, and reduces commercial friction for subsequent launches.
This model also gives Delcath strategic optionality. If partnered or acquired, the buyer inherits not a single product but a scalable, multi-indication procedure platform, akin to acquiring a surgical robot business with multiple FDA clearances. If independent, Delcath retains the ability to selectively prioritize high-margin, low-barrier indications without straining internal bandwidth.
In both paths, indication sequencing isn’t just a growth tactic, it is a defensible operating strategy that strengthens the company’s margin profile, speeds adoption, and builds a moat around its procedural footprint.
Institutional embedding and network effects: From site activation to procedural entrenchment
Delcath’s long-term success is not contingent on a blockbuster label or a singular clinical trial outcome, it hinges on a more durable and often underappreciated asset: institutional embedding. In practical terms, this means converting cancer centers not just into customers, but into permanent procedural hubs that rely on the company’s platform as part of routine practice.
Over time, the more institutions adopt, the deeper the company embeds itself within oncological workflows, generating a flywheel of procedural habit, network reinforcement, and operational dependence.
This strategy has already begun to take hold. Delcath’s mUM launch has targeted major academic centers and leading interventional oncology programs, prioritizing depth over breadth. These institutions often act as procedural opinion leaders, setting adoption standards, training junior clinicians, and publishing real-world data that shapes clinical consensus.
Once trained and reimbursed, these centers have high repeat-procedure potential, particularly for indications requiring ongoing regional control (as with mUM and CRLM). This procedural repeatability allows Delcath to turn individual sites into high-volume nodes, with robust visibility on revenue per center and growing leverage on fixed SG&A investments.
What emerges is not a transactional relationship, but a network architecture. Each center activated increases the value of the overall platform, for Delcath, for physicians, and for patients. Physicians become more proficient and efficient over time, reducing training and procedure time.
Hospitals amortize upfront infrastructure and staff training, increasing return on capital and internal justification for broader indication use. Payers observe greater procedural consistency and clinical benefit, strengthening the case for sustained reimbursement. These effects compound with each site, making the platform more defensible and reducing the likelihood of displacement by competing technologies.
This is not a static relationship. Delcath actively supports procedural adoption with a field team that offers on-site clinical guidance, reimbursement assistance, supply logistics, and patient navigation tools.
These touchpoints deepen dependence and reinforce Delcath’s position as not just a device provider, but a procedural partner. In the long term, this network becomes self-perpetuating: sites generate data, data supports broader adoption, and adoption attracts more clinicians to train, publish, and advocate. The entire procedural category thus becomes institutionally self-reinforcing.
From an investor’s perspective, this creates a powerful moat. As with surgical robotics or catheter-based interventions, once procedural infrastructure is in place, and once payers, providers, and physicians are all aligned around its clinical and economic value, the barriers to replacement become formidable. Delcath is not just seeking market share; it is seeking procedural ownership.
The network effects from site activation and physician training are not fluffy intangibles. They are tangible strategic assets, quietly compounding into long-term value.
Global expansion and non-U.S. leverage: A scalable opportunity with structural tailwinds
Delcath’s commercial runway is not limited to the U.S. healthcare system. In fact, one of the company’s most structurally underappreciated advantages is its ability to scale globally without proportional cost or risk, due to the procedural nature of its platform and the preexisting clinical precedent in European markets.
While early revenue traction is expected to be U.S.-dominated due to the formal launch of HEPZATO KIT for mUM, the real strategic tailwind lies in Europe and other international regions, where Delcath already has regulatory experience, clinician familiarity, and pricing benchmarks from earlier versions of the platform.
Delcath's CHEMOSAT system, a predecessor to the current HEPZATO KIT, was granted CE Mark in Europe more than a decade ago and used in real-world clinical settings across Germany, the U.K., France, and Italy. Although it lacked formal reimbursement in most countries, this legacy footprint gave Delcath a valuable early signal: interventional oncologists are receptive to the concept of regionally isolated liver perfusion, and the procedural workflow can be implemented in non-U.S. institutions.
This matters greatly for future scale. Unlike many oncology companies that must start from scratch internationally, Delcath enters European expansion with clinical relationships, procedural familiarity, and existing data infrastructure already in place.
As of 2024, the company is actively pursuing targeted commercialization in Germany, the Netherlands, Austria, and select Nordic countries, all of which have (1) centralized cancer centers, (2) high procedural throughput, and (3) pathways for device-procedure reimbursement under DRG-like frameworks.
Importantly, the procedural model fits well within European public healthcare systems because it consolidates treatment into single-site interventions with defined costs and outcomes, making it easier to evaluate and budget for than diffuse systemic regimens. Delcath’s support model, including physician training, regulatory filings, and field-based procedure support, can be extended across these regions with minimal incremental fixed cost, allowing the company to achieve geographic leverage without duplicating its U.S. SG&A footprint.
Looking longer term, expansion beyond Europe is also plausible. In Asia, particularly Japan and South Korea, interventional oncology is a well-established specialty with highly concentrated patient volumes at academic centers, structurally similar to the European market.
The company’s procedural IP, once established in multiple major regulatory environments, would give it a pathway to pursue market entry via local partners or licensing structures. This kind of geographic arbitrage, exporting an increasingly standardized and systematized treatment method, is what gives the platform global procedural elasticity.
From a financial perspective, global expansion increases the platform’s surface area without linear increases in OPEX. From a strategic perspective, it diversifies revenue, creates optionality for partnerships, and embeds the company across healthcare systems with varying policy, pricing, and risk profiles. In sum, Delcath’s non-U.S. strategy is not a distant bolt-on, it is a natural extension of the procedural business model, and one that reinforces the thesis of scalable, embedded hepatic oncology infrastructure.
15. Opinion & Target View
Delcath isn’t a mUM drug story, it’s a platform inflection hiding in plain sight, priced like a one-off product.
Delcath Systems sits at a rare strategic intersection: clinically de-risked, procedurally validated, and structurally mispriced. While the market still treats it like a niche therapeutic launch, what’s unfolding is a durable platform story, one that mirrors the early signals of category-defining interventional franchises. This section distills the core thesis and outlines the investment logic behind a long-duration, asymmetric bet.
A platform mispriced as a product story
Delcath Systems presents one of the clearest examples of structural asymmetry in the current small-cap biotech landscape, a commercial-stage company with a validated product, regulatory clearance in a rare cancer, a highly specific procedural advantage, and a defined path to platform expansion across multiple high-prevalence solid tumors.
This is not a speculative preclinical biotech hinging on binary readouts, nor is it a fully scaled oncology giant with priced-in expectations. Rather, Delcath sits in a transitional space, where near-term commercial execution will either unlock significant platform value or reintroduce operational drag. For long-term investors, this liminality is precisely where the most compelling upside lives.
Not a revenue ramp - a platform inflection
From an investor lens, Delcath is not about quarter-by-quarter revenue ramp or whether it beats consensus on mUM patient starts. It is about whether the company can achieve procedural embeddedness in a treatment niche with few alternatives, and whether the platform logic, that isolating liver perfusion can be used in CRLM, ICC, and potentially other liver-dominant malignancies, holds and scales.
That is a binary of a different sort: not one trial result, but an operational execution arc. Investors who understand procedural market building (e.g., Intuitive Surgical, Shockwave, TAVR in its early days) will recognize the signs, hospital adoption curves, KOL-driven uptake, reimbursement integration, and follow-on indication momentum.
Three operational questions that matter
The investor case does not hinge on near-term profitability. It hinges on a set of operational questions:
Can Delcath consistently deliver the HEPZATO procedure across centers of excellence and maintain quality of outcomes?
Can it drive sufficient reimbursement consistency and physician economics to support widespread uptake?
Can it execute the follow-on CRLM program in a way that both clinicians and regulators view as compelling?
If the answer to those questions is "yes," then Delcath is not a $300-400M microcap, it is the nucleus of a procedural oncology platform that could support a multi-billion dollar valuation over time. That path, while not guaranteed, is significantly de-risked by the FDA approval of HEPZATO for mUM and the infrastructure the company has already begun deploying.
A thesis for long-term, high-conviction investors
For long-term investors with a tolerance for execution risk and a time horizon that extends beyond the current commercial ramp, Delcath offers a high-conviction asymmetric bet. Not because the numbers look cheap, but because the market has yet to internalize that this is no longer a development-stage biotech. It is a procedurally anchored, revenue-generating oncology platform in the early innings of market expansion.
16. Conclusion: Why Delcath Represents a Rare Long-Duration Asymmetry in Procedural Oncology
This isn’t a drug launch, it’s the foundation of a category-defining procedural franchise, priced like a footnote.
Delcath Systems is not a traditional biotech story. It is a platform company that sits at the intersection of oncology, interventional radiology, and procedural standardization, with a commercial-stage therapy already FDA-approved, a scalable delivery mechanism, and a strategic roadmap targeting some of the most challenging and underserved liver-dominant cancers.
The core of the thesis is not about a single product or a narrow clinical readout, but about the progressive realization of procedural incumbency, a model that, if executed well, tends to generate compounding optionality over long time horizons.
This thesis has intentionally avoided conventional valuation modeling. That is by design. Delcath is not suited to short-term price targets or DCF-driven precision. It is better understood through strategic sequencing and inflection-stage transitions, as a company moving from regulatory validation to commercial relevance, from rare-disease penetration to platform expansion, and from cost-heavy early deployment to operating leverage and embedded growth. Each of these transitions is risky. But each also represents a force-multiplier if successfully executed.
What distinguishes Delcath is not only the differentiated delivery of its HEPZATO KIT, but the clinical logic and treatment niche it is built to dominate: liver-targeted chemotherapy in cancers where the liver is the key site of morbidity and mortality. These are patients with few other options, and physicians with few procedural tools. By building a platform that gives interventional oncologists a repeatable, reimbursable, and clinically defensible procedure, Delcath positions itself not as a single-product biotech but as a procedural platform company with durable reach.
Long-term investors looking for high-asymmetry, inflection-stage companies will recognize the setup: clinical de-risking, modest revenue traction, ongoing investment, misunderstood or under-modeled market size, and a runway of potential indication expansion. The key variable is execution, not whether the science works, but whether the company can systematically build a treatment paradigm around it.
If it does, the returns will not be linear. They will be compounding. Delcath is an early-stage procedural oncology platform with rare characteristics: scientific grounding, commercial visibility, strategic optionality, and structural scarcity. For those who can look past the current GAAP losses and near-term volatility, the long-term opportunity is substantial.
You’re not betting on the next biotech catalyst.
You’re not chasing quarterly beats or sentiment swings.
You’re securing a stake in the procedural backbone of liver-directed oncology.
17. Acknowledgements & Disclaimer
This thesis is based on personal analysis and publicly available information from a range of sources, including company filings, industry reports, and public statements. It represents my own interpretation of the data and broader market context. Nothing contained herein should be construed as investment advice or a recommendation to buy or sell any security.
Important note: This analysis is purely educational and reflects my personal views. It is not financial advice or an investment recommendation. Always do your own due diligence or consult with a licensed professional before making investment decisions.
Thanks again for reading, and as always, stay curious and long-term oriented.
Sincerely,
A.C.L.